The painting above shows acute tubular injury with numerous intracytoplasmic inclusions within proximal tubular epithelium, which can be seen in lysozyme-associated nephropathy. Lysozyme-induced nephropathy is a rare cause of acute tubular injury seen in patients with hematologic malignancies, most commonly with chronic myelomonocytic leukemia (CMML) and acute monocytic leukemia (Santoriello et al, 2017). Malignancy associated overproduction of lysozyme can lead to acute kidney injury and proteinuria. Lysozyme is freely filtered by the glomerulus and reabsorbed by the proximal tubular epithelium. While this happens at a low level physiologically, over-reabsorption of lysozyme induces toxic acute tubular injury.
Lysozyme-induced nephropathy is one differential diagnosis to consider when determining potential etiologies of acute renal failure in patients with hematologic neoplasms and may be under-recognized (Cossey et al, 2013). Acute kidney injury (AKI) affects 1/3 of patients with CMML, causing significant morbidity (Santoriello et al, 2017). Other sources of AKI in this setting include volume depletion, tumor lysis syndrome, nephrotoxicity related to chemotherapy agents, intravascular leukostasis, or leukemic infiltration of the kidney parenchyma. A clinical clue to lysozyme-associated nephropathy in these patients, although non-specific, is increased urine total protein compared to total urine albumin. This protein gap is caused by lysozymuria. Urine (and serum) lysozyme levels, while they would be elevated, are non-specific and can occur in many other conditions, including sarcoidosis, macrophage activation syndrome, carcinomatosis, typhoid fever, cadmium poisoning, Fanconi syndrome, Lowe’s syndrome, multiple myeloma, plasma cell leukemia, Hodgkin lymphoma, reticulum cell sarcoma, and others (Hillen et al, 2018). Since the potential etiologies for AKI in patients with hematologic malignancies is broad, a kidney biopsy may be required for diagnosis.
On a kidney biopsy, there are numerous hypereosinophilic granules within proximal tubular epithelium, which are weakly PAS positive and non-agyrophilic on silver stains in lysozyme-induced nephropathy. Non-agryrophilia helps distinguish these inclusions from protein resorption droplets that occur with albuminuria. An immunohistochemical stain for lysozyme can be used to highlight lysozyme accumulation within the proximal tubular epithelium. This should be compared to a control kidney biopsy (without acute tubular injury) in parallel, as low level lysozyme staining is normal within proximal tubules. The ultrastructural appearance by electron microscopy is that of numerous autophagolysosomes, which are polygonal and electron-dense intracytoplasmic structures.
Light and electron microscopic images from a case of lysozyme-induced nephropathy are shown below. This entity should be considered as one of many potential etiologies of AKI in patients with hematologic malignancies.
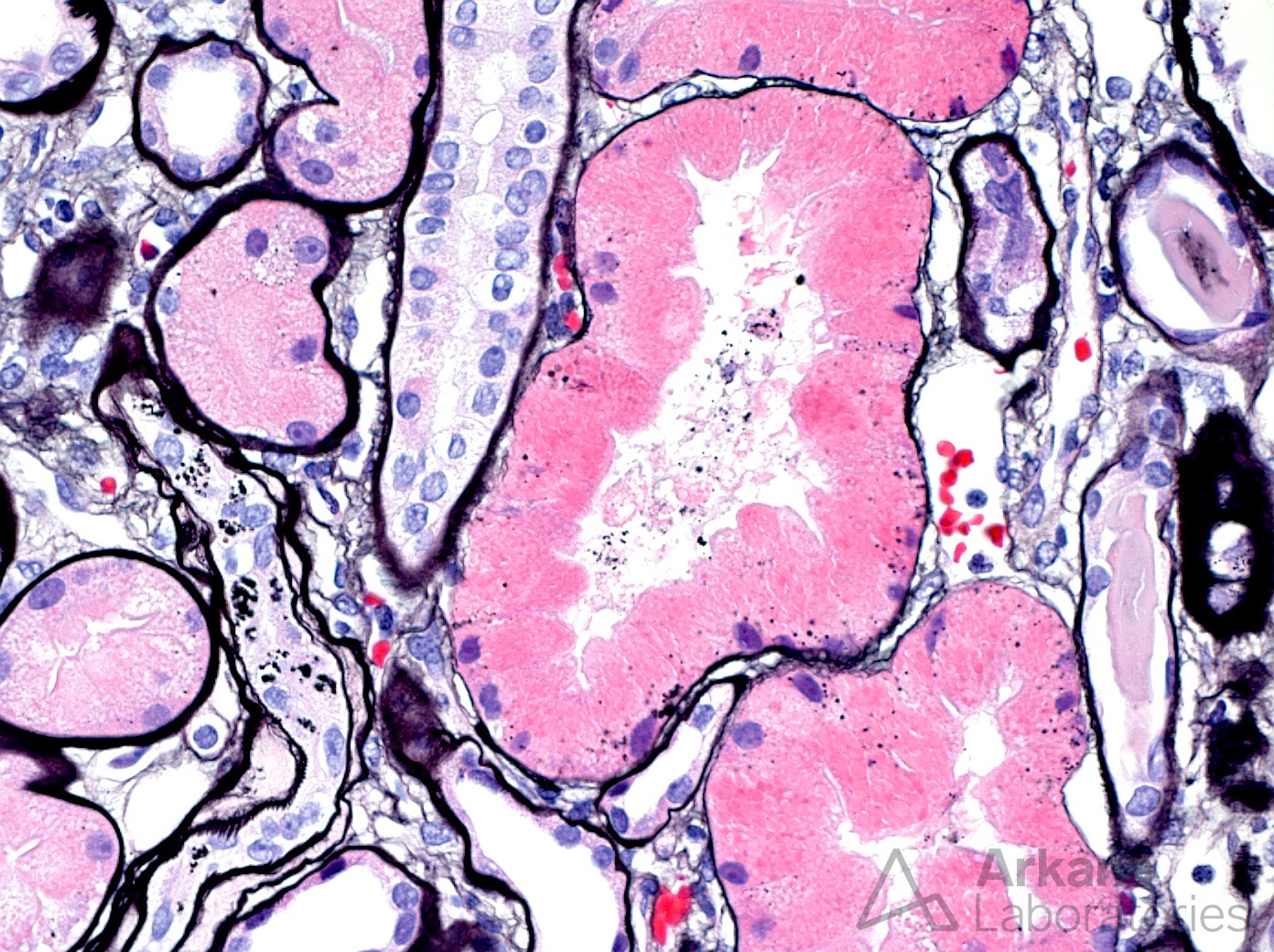
JMS stain showing non-agyrophilic granularity to proximal tubules.
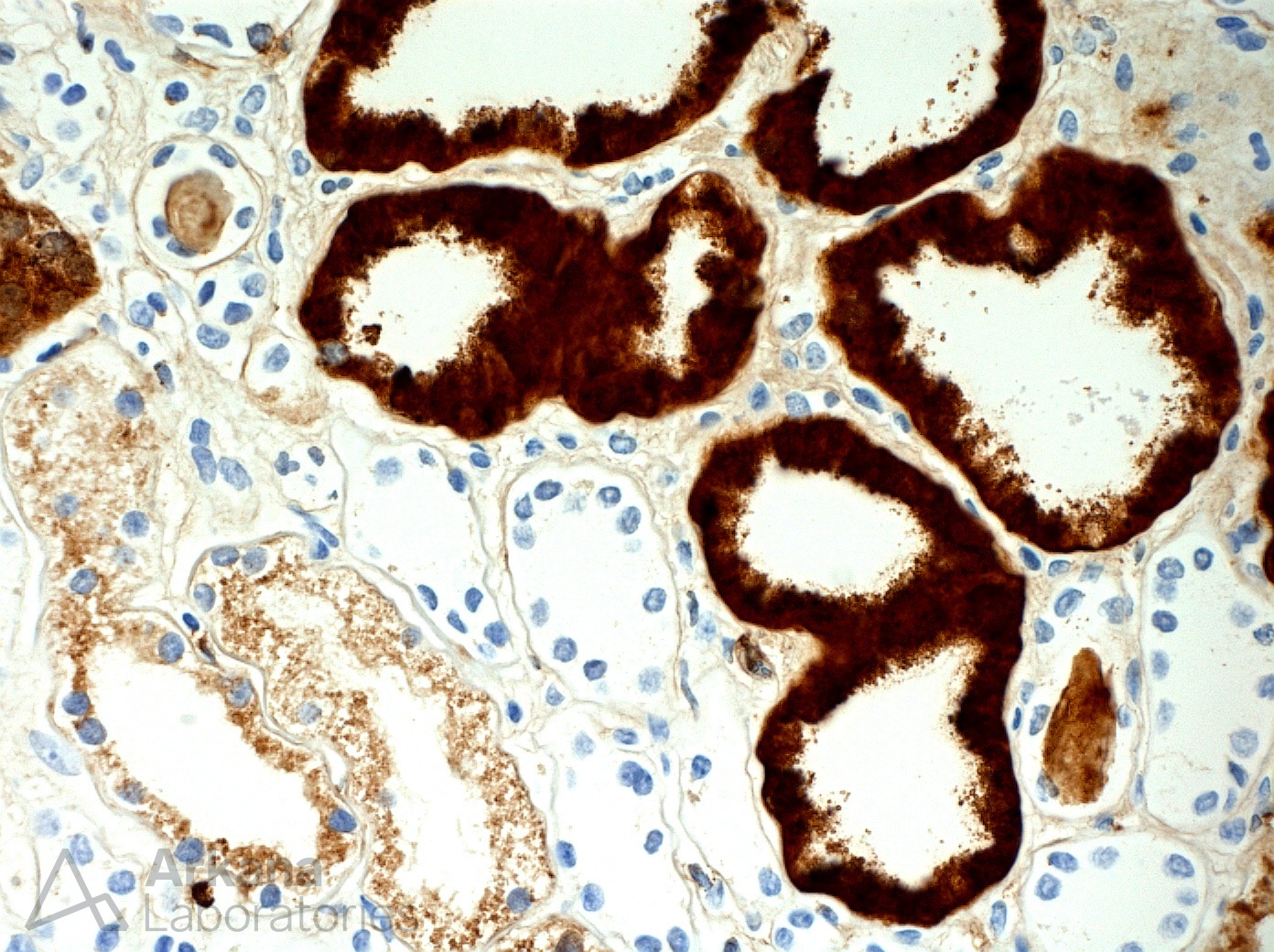
Lysozyme immunohistochemistry showing strong expression within proximal tubules.
References:
Cossey LN, Larsen CP. Lysozyme-Induced Acute Tubular Injury: An Under-Recognized Cause of Acute Renal Failure. Poster Presentation. American Society of Nephrology 2013 annual meeting, Atlanta, GA.
Hillen JM, Raemaekers JM, Steenberger EJ, Wetzels JFM, Verhave JC. Progressive kidney failure in chronic myelomonocytic leukemia: don’t forget about lysozyme damage. The Netherlands Journal of Medicine 2018 Nov; 76 (9): 407-411.
Santoriello D, Andal LM, Cox R, D’ Agati VD, Markowitz GS. Lysozyme-induced nephropathy. Kidney International Reports 2017 Jan; 2 (1): 84-88.
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.


