The patient is a 45-year-old IV drug user who presents with fevers, chills, hematuria, and fatigue. On laboratory testing, he was found to have a creatinine of 4.8. Complement levels are depressed and blood cultures are positive. A transesophageal echocardiogram confirmed the presence of endocarditis. A kidney biopsy was obtained due to hematuria.
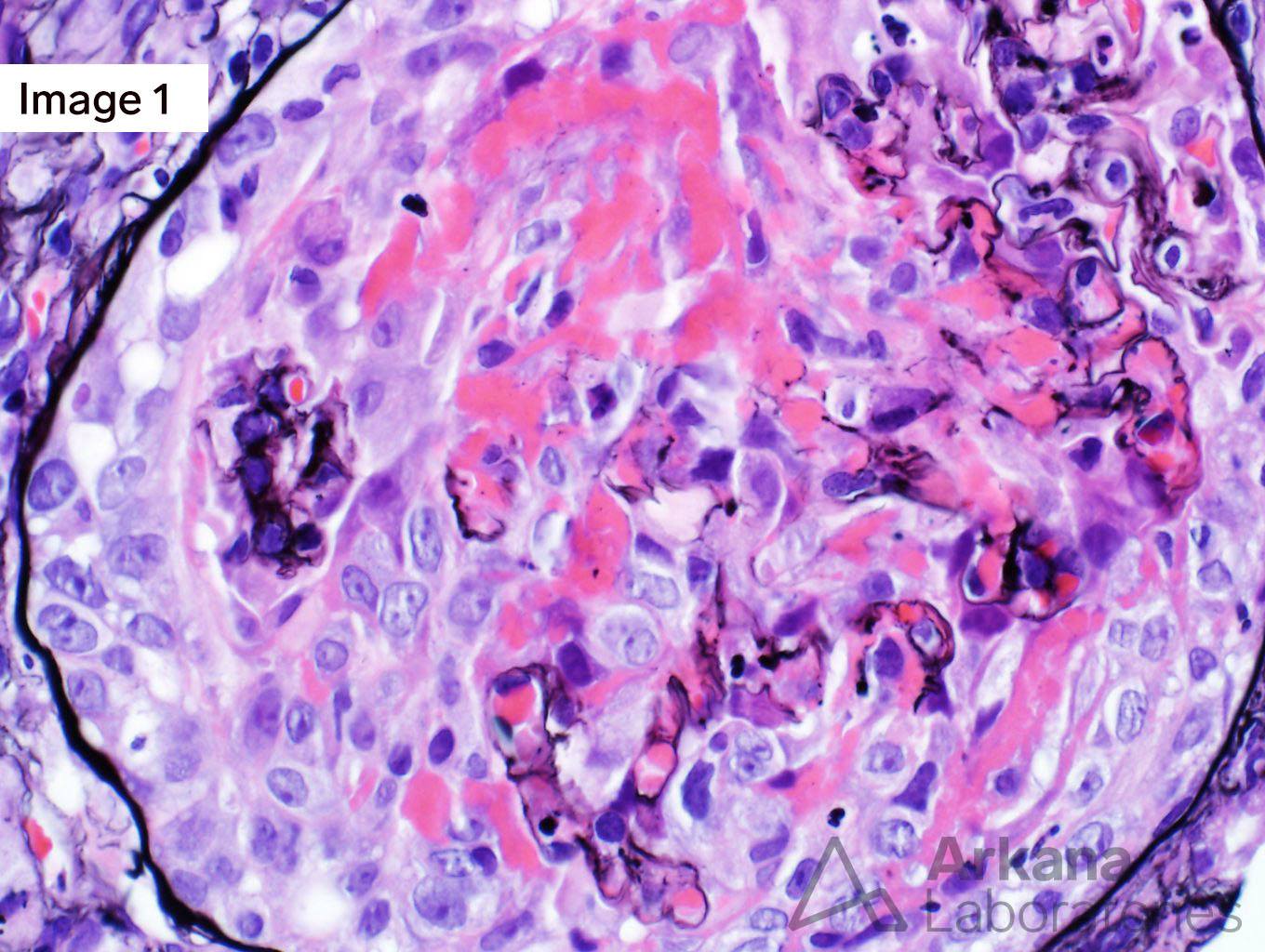
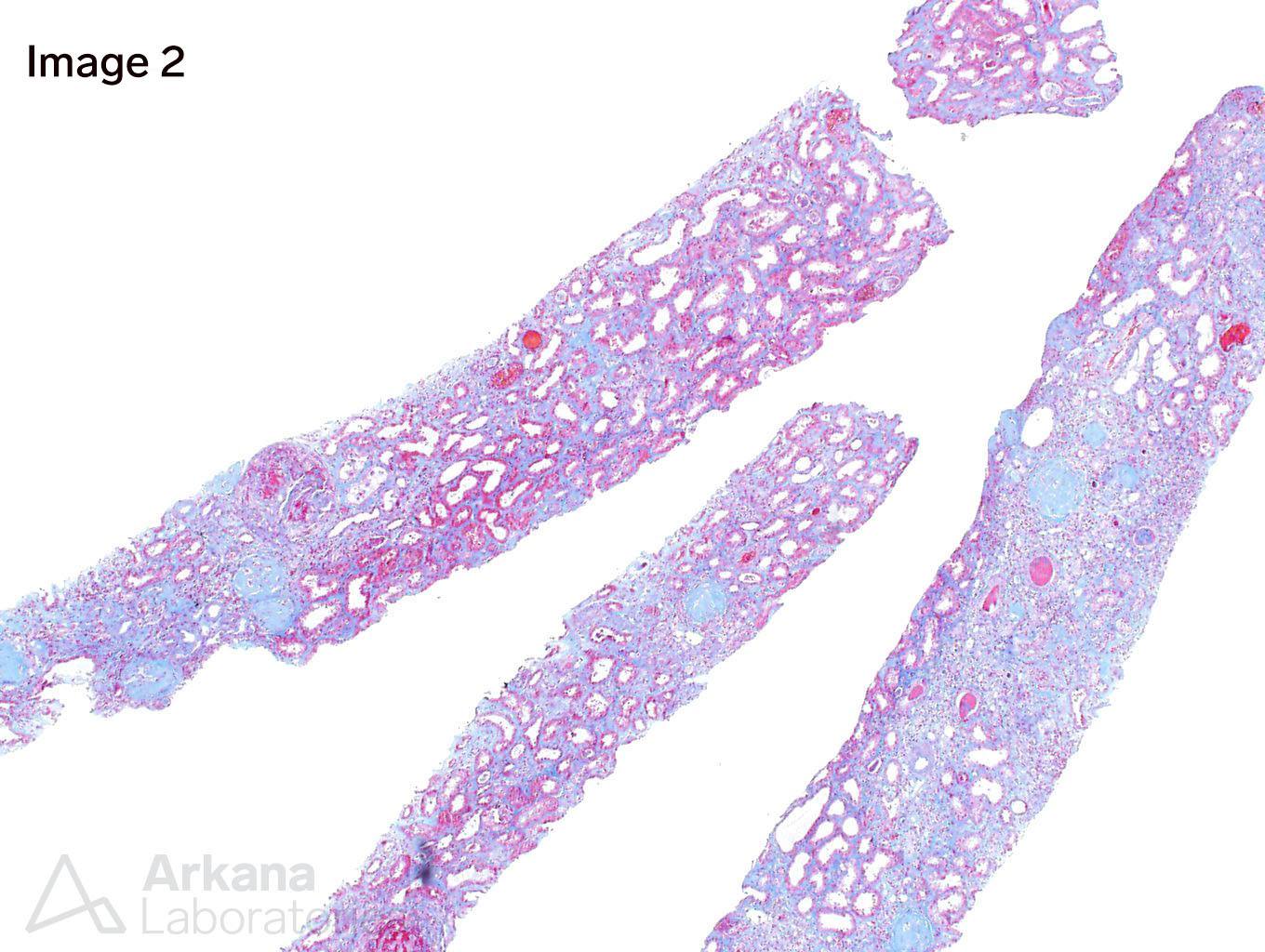
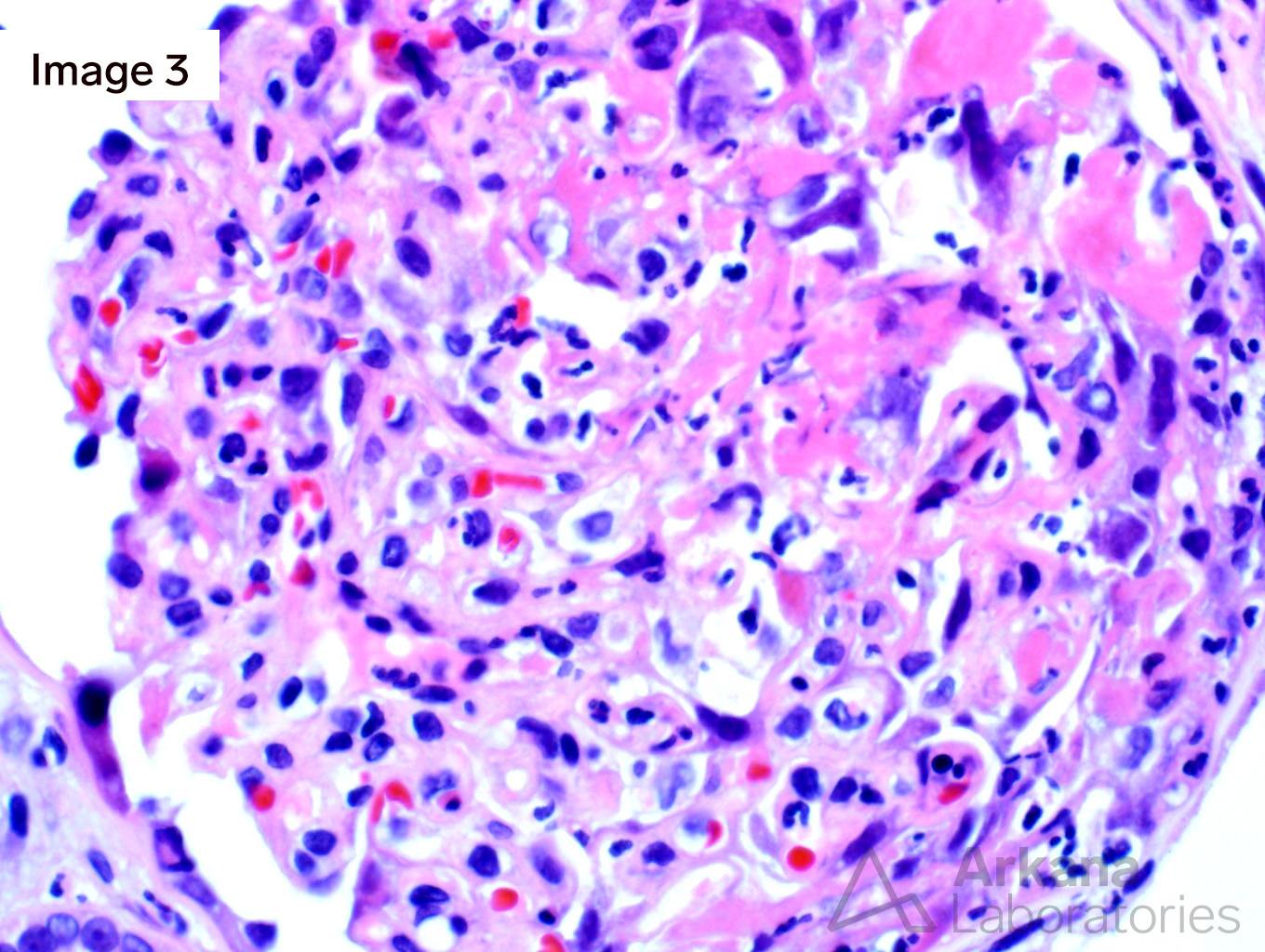
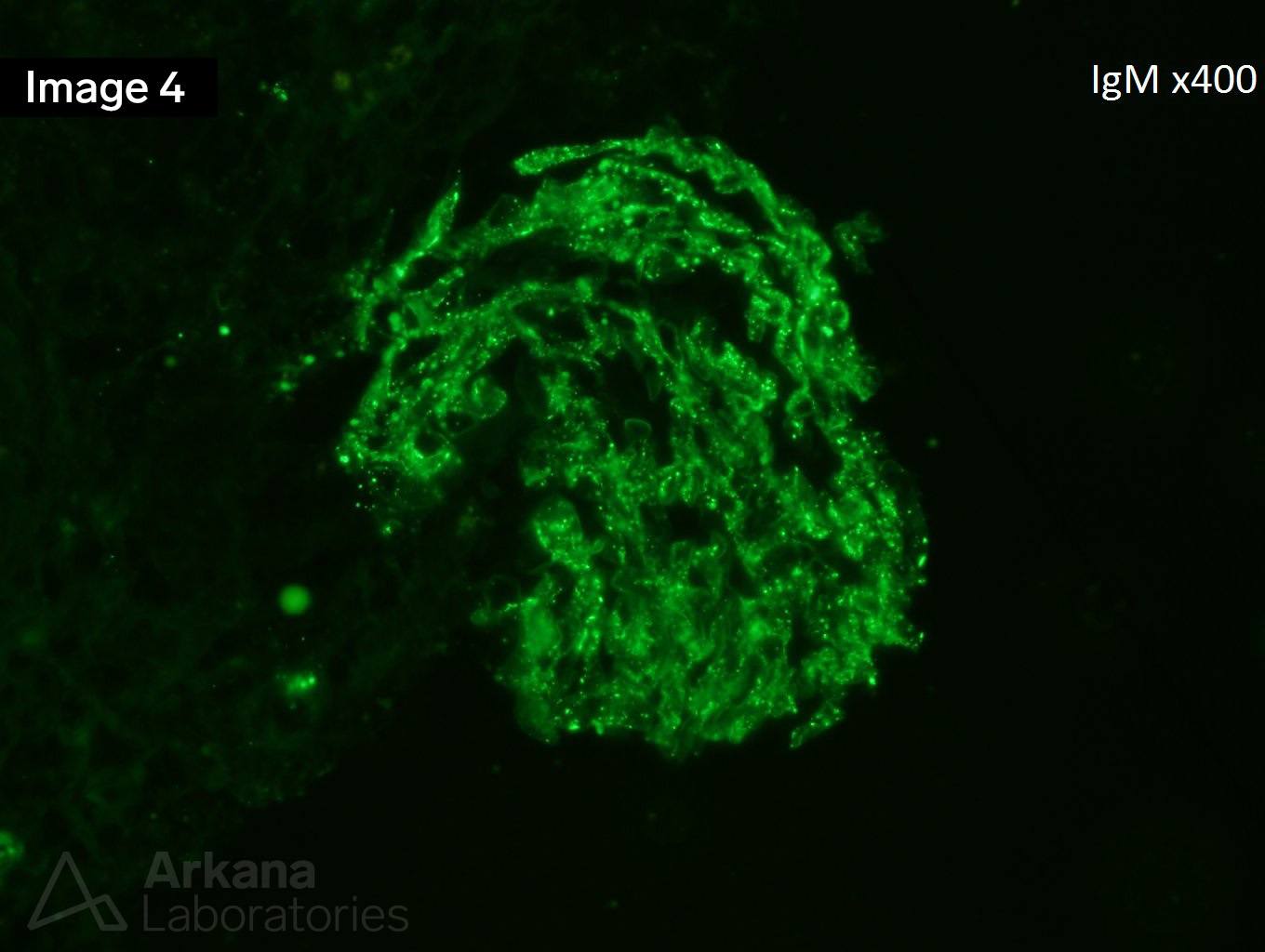
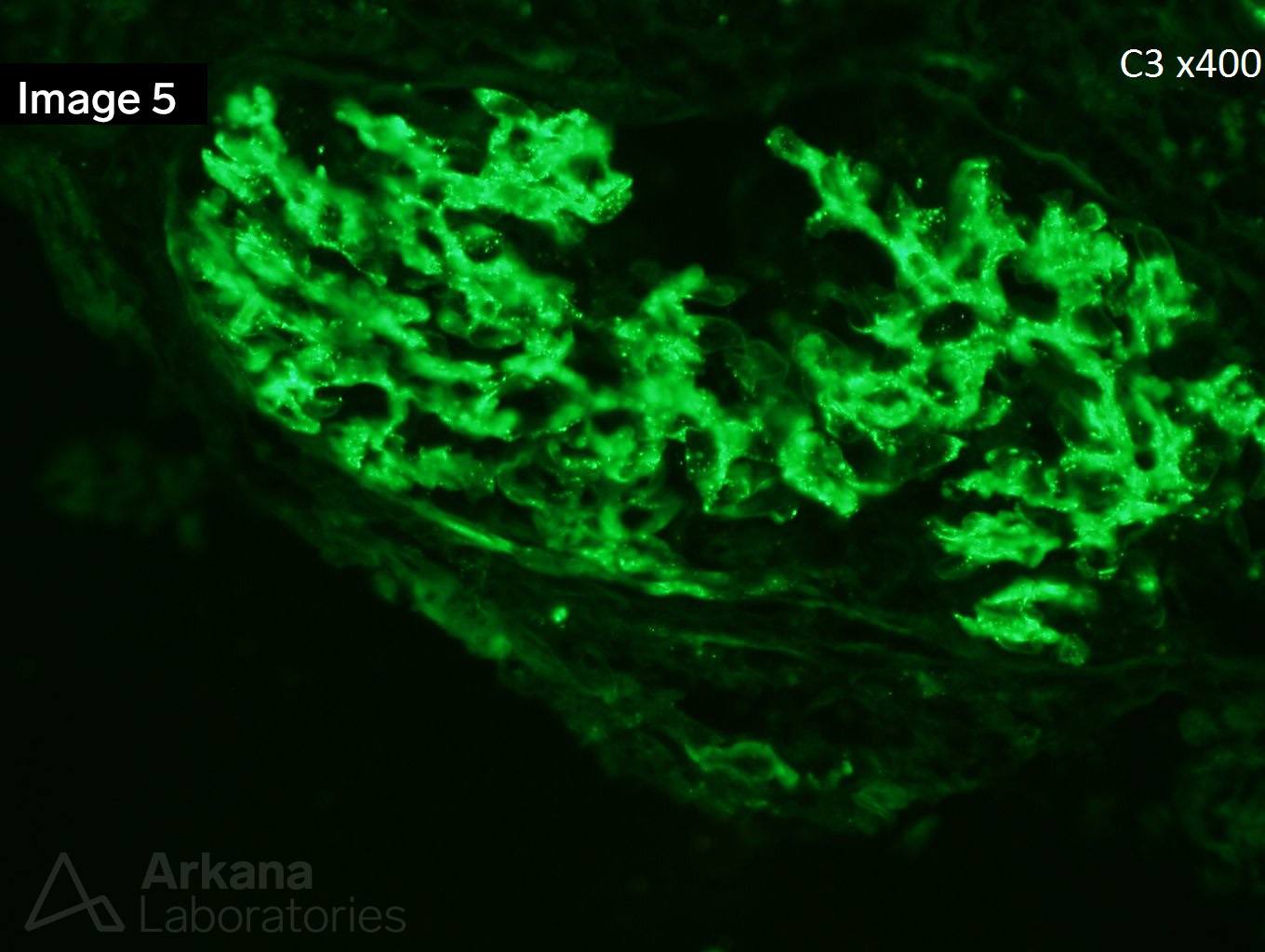
Image 1 and Image 3 shows circumferential cellular crescents with fibrinoid necrosis (many intracapillary neutrophils are seen in image 3). Figure 2 shows severe interstitial fibrosis. Images 4 and 5 show immunofluorescence staining with IgM and C3, respectively.
This is a case of endocarditis-associated glomerulonephritis with diffuse crescent formation. Contrary to infection-associated glomerulonephritis in general, the most common pattern of glomerular injury in infective endocarditis-associated glomerulonephritis is necrotizing and crescentic glomerulonephritis. ANCA-associated glomerulonephritis enters the differential and up to 28% of endocarditis-associated glomerulonephritis has positive ANCA serology. The strong staining for both IgM and C3 is more commonly seen in endocarditis-associated glomerulonephritis as compared to pauci-immune mediated glomerulonephritis. The most common comorbidities associated with endocarditis-associated glomerulonephritis are cardiac valve disease, intravenous drug use, hepatitis C, and diabetes. Treatment relies on antibiotics plus immunosuppressive therapy and in some cases surgical replacement of cardiac valves.
https://www.ncbi.nlm.nih.gov/pubmed/25607109
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.