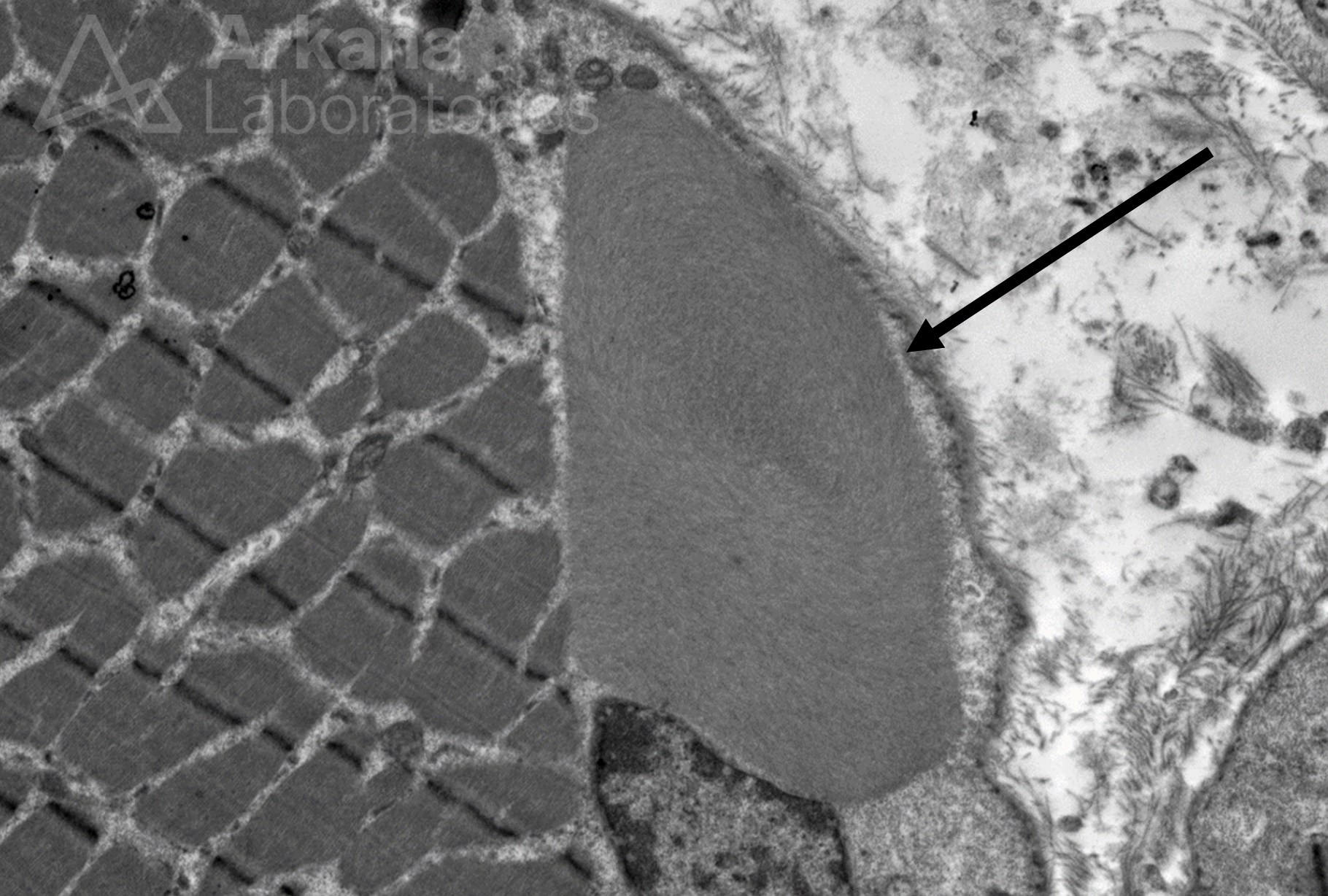
A 75-year-old female patient presents with proximal muscle weakness, characteristic skin rash, and dysphagia. Laboratory workup reveals elevated CPK 3500. A skeletal muscle biopsy is performed from the right thigh and shows findings diagnostic of dermatomyositis. The muscle biopsy also shows rare vacuolated muscle fibers with Modified Gomori Trichrome stain. Electron microscopy is pursued. What is the ultrastructural finding seen in the electron photomicrograph shown below?
Answer: The electron photomicrograph shows a Fibrous (filamentous) body, which is a nonspecific finding that may be seen in a variety of clinical settings, including inflammatory myopathies, such as dermatomyositis, as well as in a normal skeletal muscle. Fibrous bodies are thought to be of myofibrillar origin and are composed of tightly packed actin-like filaments. Vacuole-type structures have been reported in biopsy-proven dermatomyositis. To learn more about the ultrastructural findings in muscle biopsies, see the references below.
Why were the other answers incorrect?
The ultrastructural distinction between a filamentous inclusion of Inclusion Body Myositis (IBM) and a nonspecific fibrous (filamentous) body is an important one where misinterpretation could lead to a diagnostic pitfall. The inclusions of IBM may be found in the sarcoplasm and in intranuclear locations, and are composed of fibrils that typically range 16-18 nm in diameter. Rimmed vacuoles are characteristic of sporadic Inclusion Body Myositis (sIBM). The clinical vignette givens a generally incorrect pattern of muscle weakness for sIBM, and the CPK elevation is too high for a typical presentation of sIBM.
Fingerprint bodies are a rare finding. Increased numbers of fingerprint bodies have been described in the disorder “fingerprint body myopathy,” which is a muscle disorder characterized by congenital hypotonia and weakness.
Cytoplasmic bodies are a very common finding thought to be due to an abnormality of the Z line (a.k.a. Z disc). These structures are dark with Modified Gomori Trichrome preparation by light microscopy. Electron microscopy yields a dense circular to oviod center with a peripheral zone of radiating fine filaments. “Cytoplasmic body myopathy” was described several years ago, and is now believed to be due to a mutation in the skeletal α-actin gene.
References:
Curless RG, Payne CM, Brinner FM. Fingerprint body myopathy: a report of twins. Dev Med Child Neurol. 1978 Dec;20(6):793-8. PMID: 729932.
Donkervoort S, Chan SHS, Hayes LH, et al. Cytoplasmic body pathology in severe ACTA1-related myopathy in the absence of typical nemaline rods. Neuromuscul Disord. 2017 Jun;27(6):531-536. PMID: 28416349.
Dubowitz V, Sewry CA, Oldfors A. Ultrastructural Changes. In: Muscle Biopsy: A Practical Approach, 5th ed. Elsevier Limited. 2021: 78-139.
Fardeau M, Tomé FM, Derambure S. Familial fingerprint body myopathy. Arch Neurol. 1976 Oct;33(10):724-5. PMID: 184767.
Greenberg SA. Inclusion body myositis: clinical features and pathogenesis. Nat Rev Rheumatol. 2019 May;15(5):257-272. PMID: 30837708.
Goebel HH, Bönnemann CG. Rare Structural Myopathy Consortium. 169th ENMC International Workshop Rare Structural Congenital Myopathies 6-8 November 2009, Naarden, The Netherlands. Neuromuscul Disord. 2011 May;21(5):363-74. PMID: 21414784.
Limaye VS, Blumbergs P. The prevalence of rimmed vacuoles in biopsy-proven dermatomyositis. Muscle Nerve. 2010 Feb;41(2):288-9; 288. PMID: 20082411.
Schuelke M, Schwarz M, Stenzel W, Goebel HH. Cytoplasmic body myopathy revisited. Neuromuscul Disord. 2018 Nov;28(11):969-971. PMID: 30253894.
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.