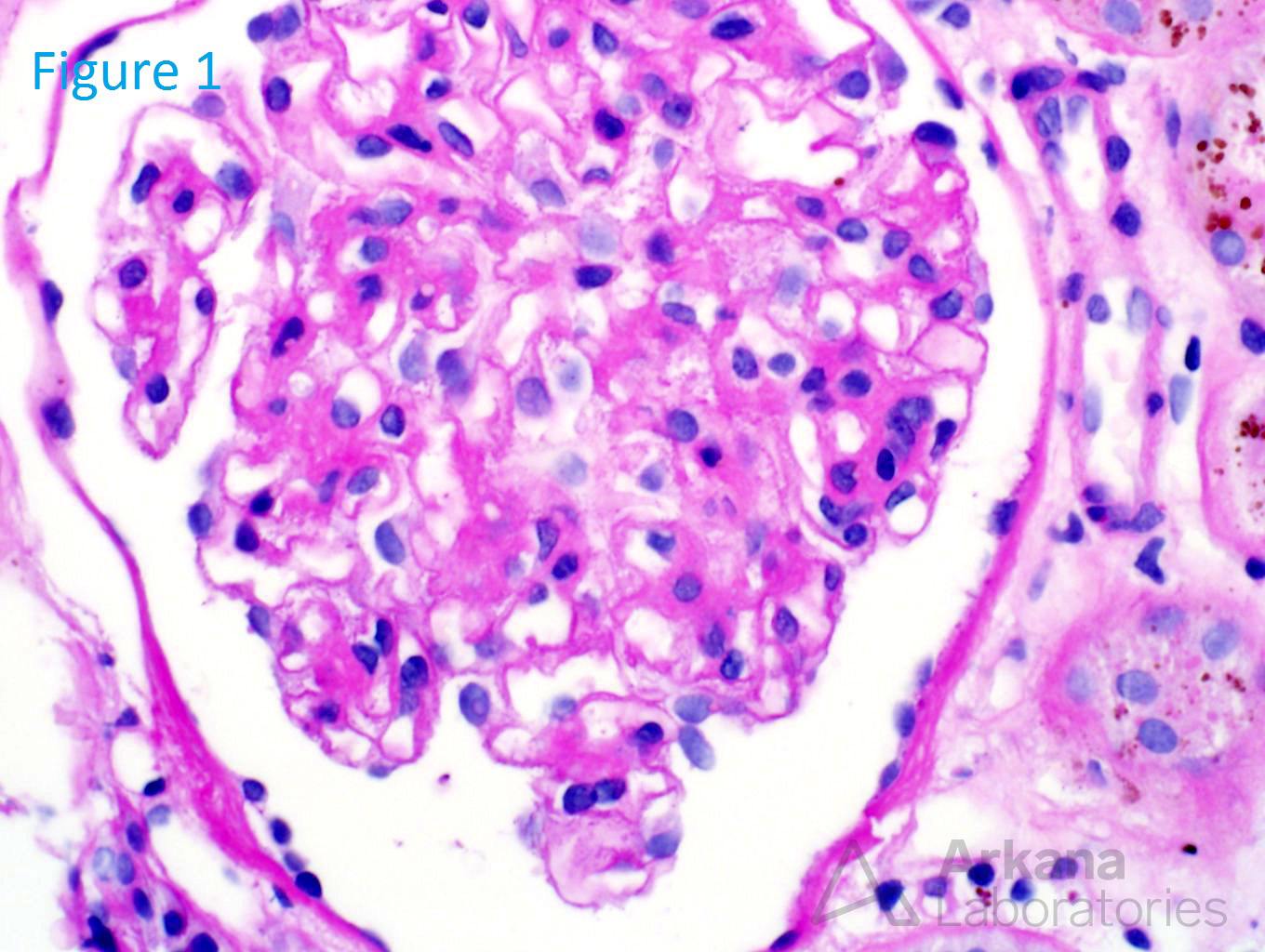
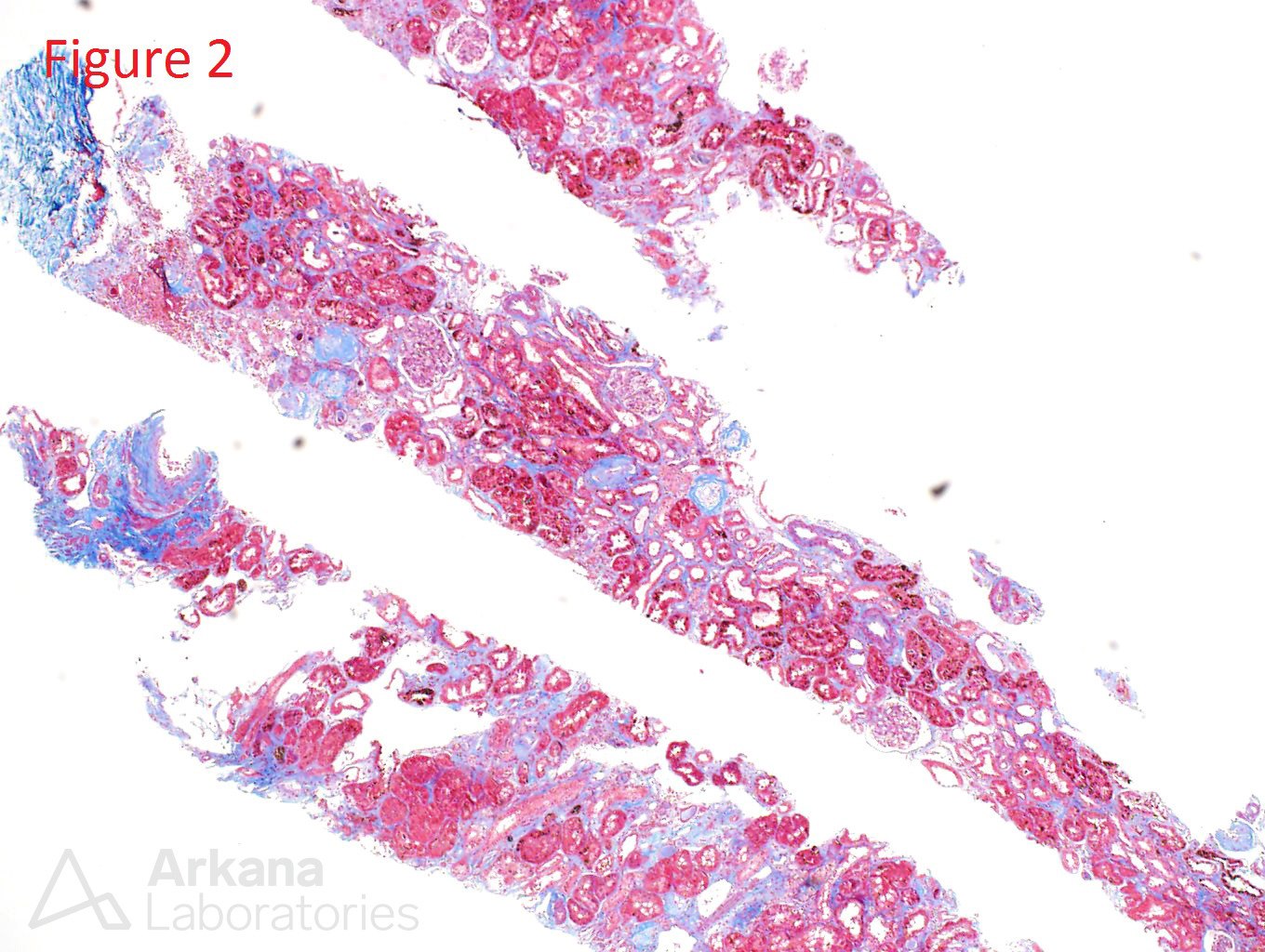
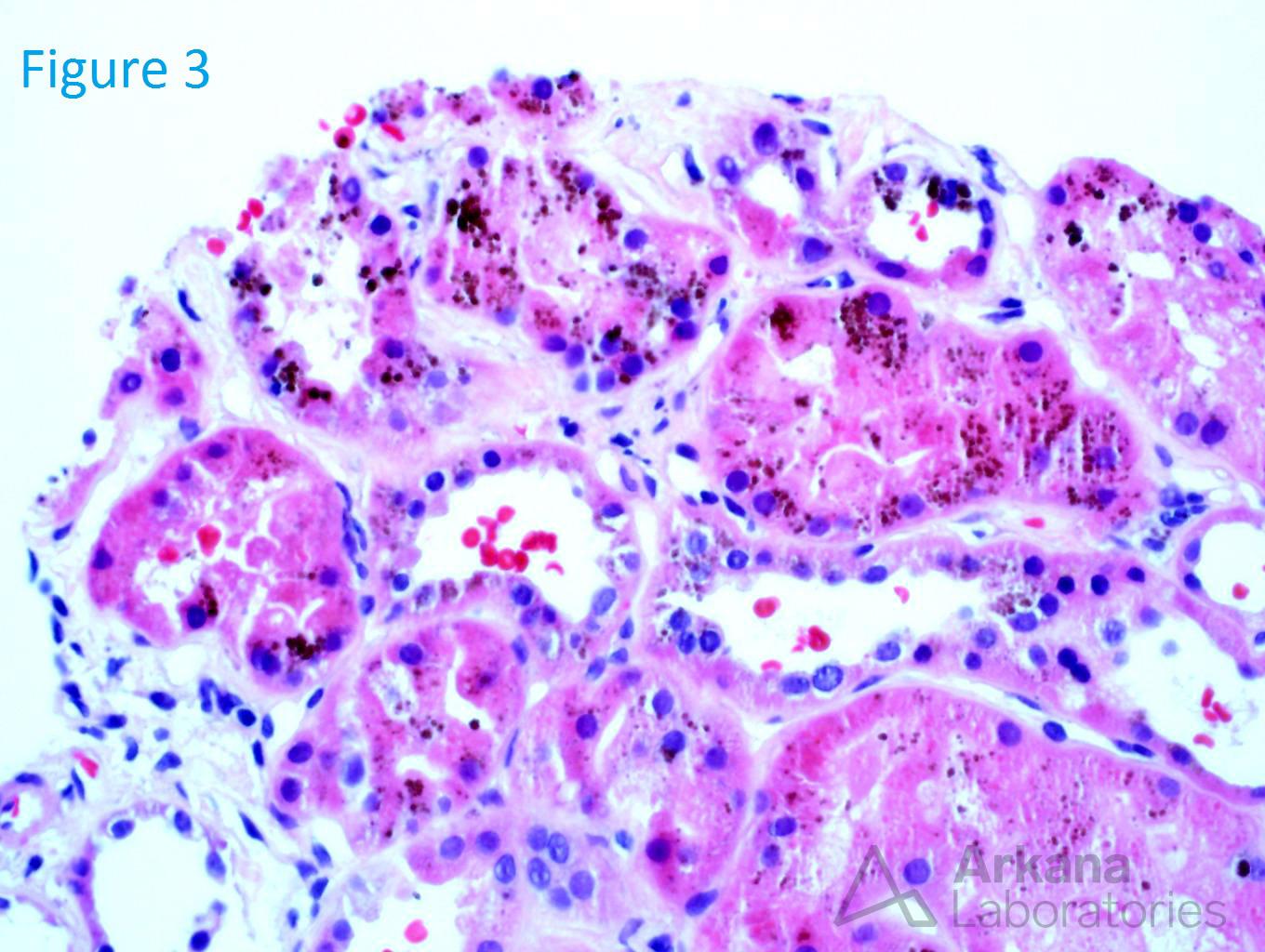
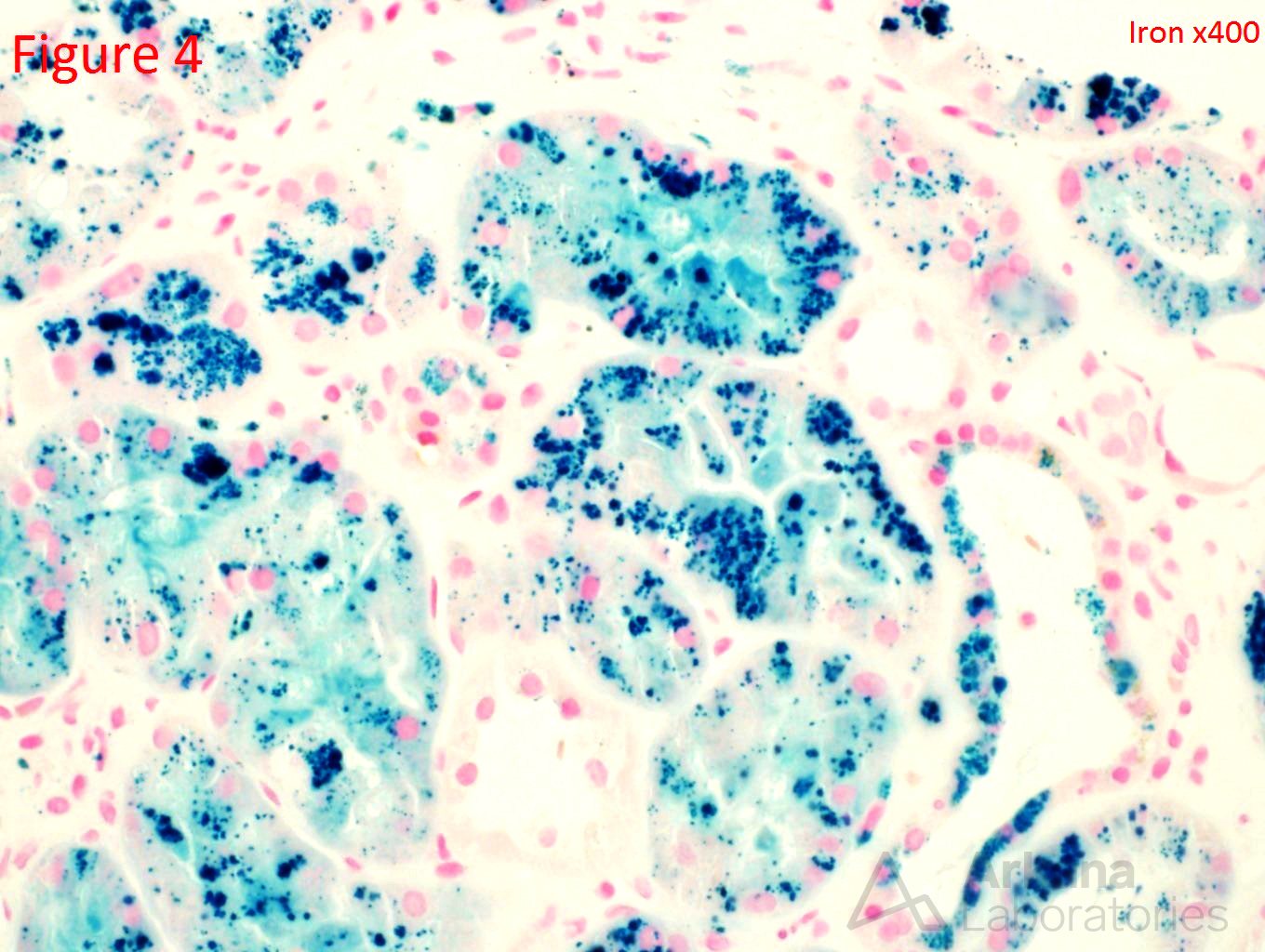
An 80-year-old white male presents with a creatinine of 4.2 (baseline 1.1) on routine lab examination. His medical history is significant for coronary artery disease, aortic valve replacement, hypertension, and peripheral artery disease. He reports no episodes of dehydration and reports that he has been in his normal state of health. Figure 1 shows a normal glomerulus. Figure 2 shows a mild interstitial fibrosis. Figure 3 shows brown spherules within the tubular epithelial cells. Figure 4 confirms the presence of iron within these brown spherules.
This is a case of renal hemosiderosis. The presence of iron deposits speaks towards intravascular hemolysis and the resorption of hemosiderin within the tubular epithelial cells. The differential includes shearing insults (prosthetic and mechanical heart valves), sickle cell disease, autoimmune etiologies, and paroxysmal nocturnal hematuria. In this patient, he had a mechanical heart valve which led to this hemolysis. The pathophysiology involving mechanical heart valves is believed to be due to altered hemodynamic shear forces at the heart valve leading to intravascular hemolysis which leads to free hemoglobin released into the circulation. The free hemoglobin rapidly binds to haptoglobin. The excess free hemoglobin which is not bound to haptoglobin is filtered freely through the glomerulus and reabsorbed by the proximal tubular cells. In the proximal tubular cells, the hemoglobin is catabolized, resulting in the release of iron in the form of hemosiderin. The hemosiderin deposited within the proximal tubular cells leads to tubular injury through oxidative stress by free radical formation and lipid peroxidation. A second mode of injury to the tubular cells is believed to be due to free hemoglobin reacting with nitric oxide to form methemoglobin and nitrate which leads to damage of endothelial cells and ischemia. Lastly, the acidic environment within the distal tubules can lead to intratubular methemoglobin cast formation which leads to intra renal obstruction and can further provoke increased uptake of toxic heme by the proximal tubules. The primary treatment of patients with severe hemolysis is repair or replacement of the prosthetic valve. Surgical revision has successfully resolved the problem in most patients. A less invasive approach is the use of antioxidant agents (N-acetylcysteine, Pentoxifylline) directed at increasing red blood cell flexibility or decreasing hydrodynamic shear forces. Propranolol has also been shown to decrease the amount of hemolysis through unknown mechanisms.
https://www.ncbi.nlm.nih.gov/pubmed/18511167
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.