Monday
Introduction into zebrafish as a model organism
Did you know? We have a state-of-the-art zebrafish facility here at Arkana Laboratories where we perform basic research with the goal of finding novel diagnostic and therapies for kidney diseases.
https://www.arkanalabs.com/research/zebrafish/


Zebrafish (Danio rerio) is a small tropical fish native to South Asia that can be purchased from your local pet shop. Indeed, the first laboratory strains were derived from several pet shops in Oregon.

Traditionally, it has been a popular vertebrate model to study developmental biology due to its rapid development outside of the uterus and transparent embryos.
https://dev.biologists.org/content/123/1/461
http://homepages.wmich.edu/~dkane1/flipbook/FlipbookMovie.mp4
Reference for both the video and the above figure: RO Karlstrom and DA Kane. A flipbook of zebrafish embryogenesis. Development (1996) 123:461.
Here’s the pubmed: https://www.ncbi.nlm.nih.gov/pubmed/9007263 for the RO Karlstrom reference
Animal models that replicate aspects of human diseases are needed in order to identify novel therapies. Due to its high fecundity, small size, fast development, and drug penetrant embryos, zebrafish bridges the gap between cell culture and the costly mammalian models.
Greater than 70% of human genes are found in zebrafish, including 82% of known disease-causing genes. Thus, it is not surprising that over the past two decades, zebrafish has become a potent model to study many aspects of human health and development. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3386812/
With the ability to perform genetic manipulation, many available genetic tools and mutants, zebrafish offers the ability to study many major organ systems, including the kidney, in the relevant context of intact vertebrates. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3386812/pdf/JCI60434.pdf
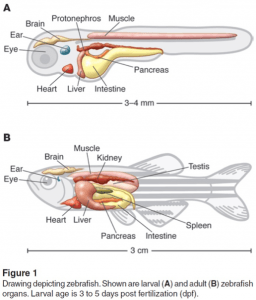
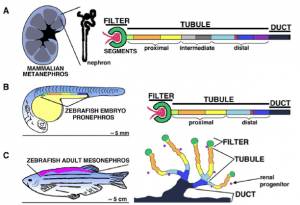
The larval zebrafish kidney (pronephros) is fully functional at 3.5 days old and is regionalized similar to the mammalian metanephros. Formation of the adult zebrafish kidney (mesonephros) occurs after the zebrafish undergo a postembryonic metamorphosis at ~10 days old. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3946610/
Tuesday
How zebrafish is being used to model and study kidney injuries
Acute kidney injury can be induced in zebrafish via injection of the aminoglycoside gentamicin, leading to tubular damage including epithelial cell flattening, thinning of the apical brush border, tubular distention, debris in the lumen, and loss of cell polarity. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3946610/
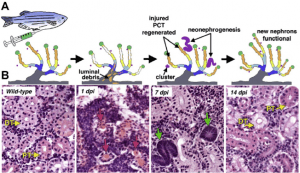
Mammals have a limited ability to heal injured nephrons. However, adult zebrafish have a robust capacity to regenerate injured nephrons within weeks of injury by expansion and differentiation of a stem cell-like population of renal progenitors. https://doi.org/10.1038/nature09669
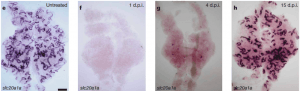
Adapted from Figure 1 from Diep, C., Ma, D., Deo, R. et al. Identification of adult nephron progenitors capable of kidney regeneration in zebrafish. Nature 470, 95–100 (2011) doi:10.1038/nature09669
Using a transgenic zebrafish line, podocyte injury can be conditionally induced, mimicking aspects of renal diseases with podocyte injury such as segmental glomerulosclerosis and membranous nephropathy. https://www.ncbi.nlm.nih.gov/pubmed/22440901 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3672345/
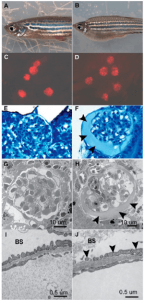
Figure 3 from Zhou and Hildebrandt. J Am Soc Nephrol. 2012 Jun;23(6):1039-47. doi:10.1681/ASN.2011080776
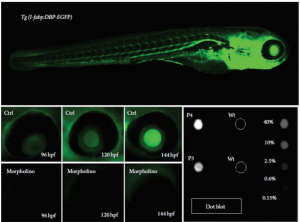
Since no albumin ortholog has been identified in zebrafish, proteinuria is modeled by monitoring the level of FITC-labeled dextran or GFP-fused to Vitamin D-binding protein (VDBP, the closest homolog to mammalian albumin) in the tubules or vasculature.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3784067/
https://www.ncbi.nlm.nih.gov/pubmed/22440901
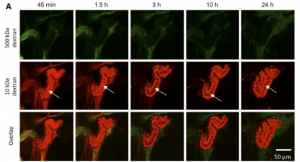
Glomerular barrier function is assessed through monitoring the levels of dextrans with different molecular weight in the circulation and in the tubules.
Wednesday
How zebrafish aids in the identification of novel therapies
Zebrafish has the potential to be a high-throughput in vivo platform for drug screening and discovery. They have a large clutch size. The embryos are small, transparent, drug penetrant, and develop outside of the uterus. https://www.nature.com/articles/nrd4627
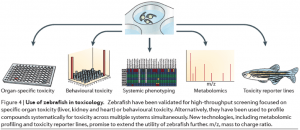
From “aquarium to bedside”: In addition to being used as an in vivo confirmation of in vitro results, researchers have identified at least 10 therapeutic compounds from zebrafish studies, some with FDA approvals for use in human. https://www.nature.com/articles/d41573-019-00165-x
Zebrafish is gaining traction in precision medicine, especially in oncology, where genetic cancer modeling is possible. Zebrafish patient-derived xenografts can also be used as avatars to assess patient’s chemosensitivity in order to guide treatment decision.
https://www.pnas.org/content/114/39/E8234
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5688753/
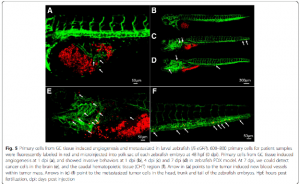
Genetic diseases can also be modeled in zebrafish. Earlier this year, a group of researchers reported a lymphatic anomaly zebrafish model, which was later used to identify a treatment that saved a patient’s life.
https://www.nature.com/articles/s41591-019-0479-2
https://www.statnews.com/2019/07/01/precision-medicine-zebrafish-helped-save-boys-life/
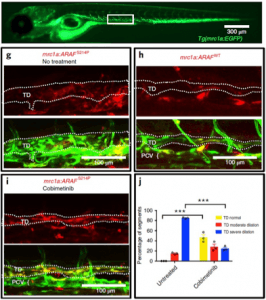
dapted from Figure 2 from Li, D., March, M.E., Gutierrez-Uzquiza, A. et al. ARAF recurrent mutation causes central conducting lymphatic anomaly treatable with a MEK inhibitor. Nat Med 25, 1116–1122 (2019) doi:10.1038/s41591-019-0479-2
Autosomal dominant tubulointerstitial kidney disease is a rare condition that often leads to ESRD and is linked to mutations in one of three genes. Pathogenesis of a newly discovered mutation was confirmed by expression of the potentially faulty gene in zebrafish. https://www.nature.com/articles/s41598-019-48014-6
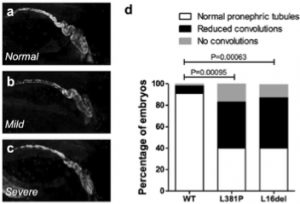
Figure 5 from Schaeffer, C., Izzi, C., Vettori, A. et al. Autosomal Dominant Tubulointerstitial Kidney Disease with Adult Onset due to a Novel Renin Mutation Mapping in the Mature Protein. Sci Rep 9, 11601 (2019) doi:10.1038/s41598-019-48014-6
Friday
Zebrafish first emerged over three decades ago as a promising animal model to better understand embryonic development in vertebrates. Recently, it has gained a foothold as a model to study human diseases and for drug discovery. Despite the vast amount of evolutionary time separating fish from humans, we share important similarities including most of our major organ systems as well as 70% of our genes. As such, many diseases can be modeled and studied in zebrafish. Unlike mammals, adult zebrafish have a nearly unparalleled regenerative capacity to replace and repair injured tissues including heart, spinal cord, and brain. Understanding how zebrafish can recover from conditions that would irreparably harm us could open the door to improved treatments.
The use of zebrafish as a model organism was pioneered to overcome some of the difficulties that plague other vertebrate model organisms such as in utero development, low offspring number, lack of genetic tractability, and high maintenance costs. Zebrafish entered the field at a time when many molecular biology techniques including mutagenesis and cloning of genes had already been developed in other model organisms. As such, these molecular techniques and other advances were quickly adapted and developed in zebrafish. Today many transgenic lines exist that enable one to look at a specific organ or a subset cell population. Many mutants with define phenotypes have also been created and characterized, allowing one to better understand the molecular mechanism underlying the genetic anomaly. The advent of CRISPR technology allows genome editing with the precision that one could only have imagined to be possible just less than 10 years ago. Finally, the advent of ever more sophisticated imaging techniques have made the zebrafish, which are transparent as larvae, a more popular research subject.
When I entered graduate school in 2008, behavioral studies and small molecule screening in zebrafish were at very early stages. Today, however, mechanisms underlying behavioral aspects of many complex neurological diseases have been elucidated using this model organism and many therapeutic compounds have already been identified from studies in zebrafish. With increasing demands to reduce our reliance on research animals, a vertebrate like the zebrafish can potentially bridge the gap between cell culture models (which lack the complexity required to study many diseases) and more traditional animal models like mice and rabbits. Furthermore, zebrafish is the only vertebrate model organism that has the potential for high-throughput screening for toxic and therapeutic compounds. Combining this with precise genome editing, it is not so hard to imagine that zebrafish could play a large role in personalized and precision medicine.
The use of zebrafish in research offers promise to better understand and treat renal diseases. While much pioneering work has been done in order to study the kidney in zebrafish (some of which were discussed earlier in the week), there is still much to do and refine. Zebrafish allow us to study the kidney in different contexts: the embryonic zebrafish kidney (pronephros) allows one to study a simpler structure; the adult zebrafish kidney (metanephros) allows one to study more complex phenotypes. I am beyond excited to be a part of a group that will work toward developing zebrafish models that will aid in identifying novel diagnostics and therapies for kidney diseases.
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.



