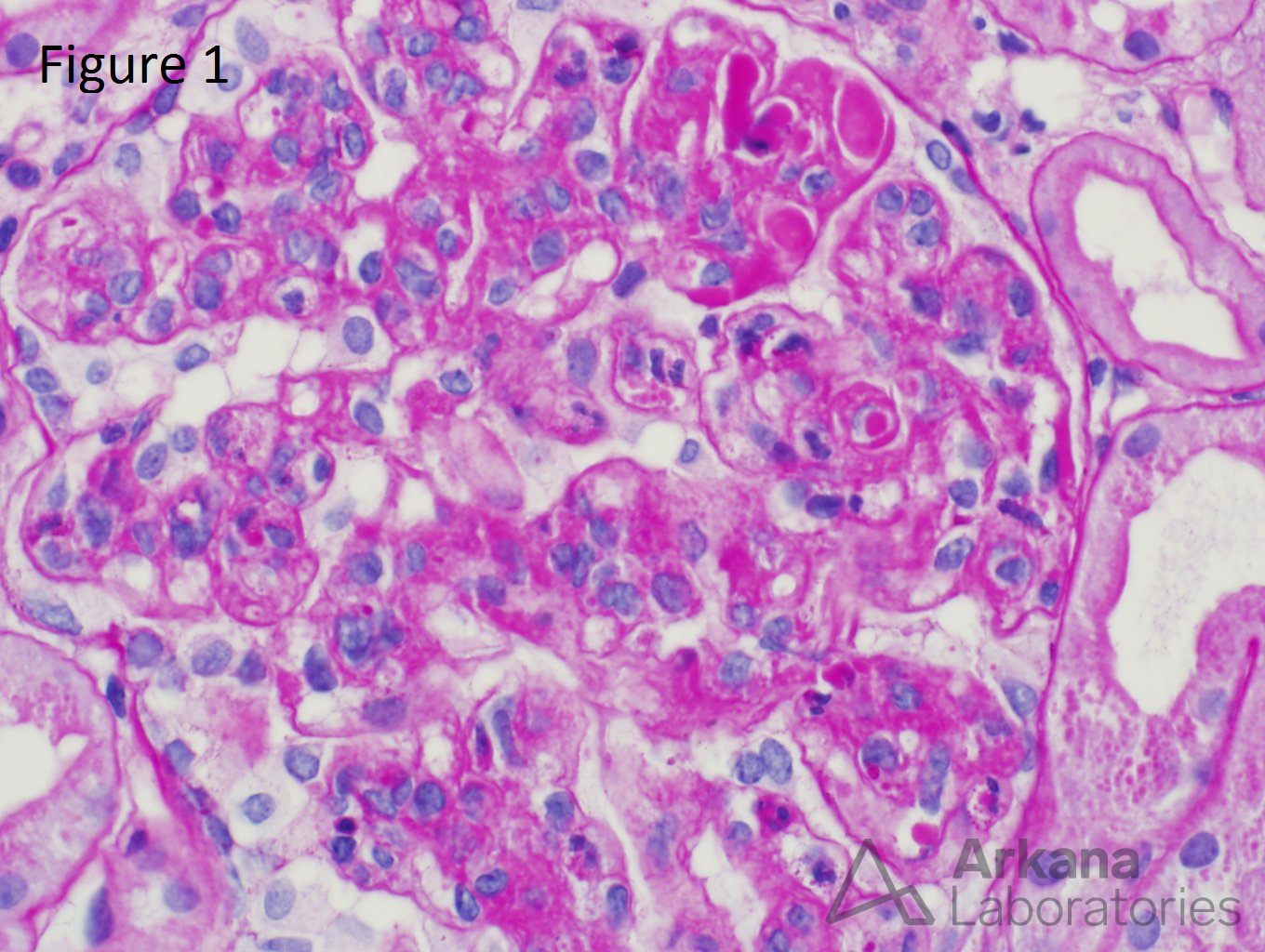
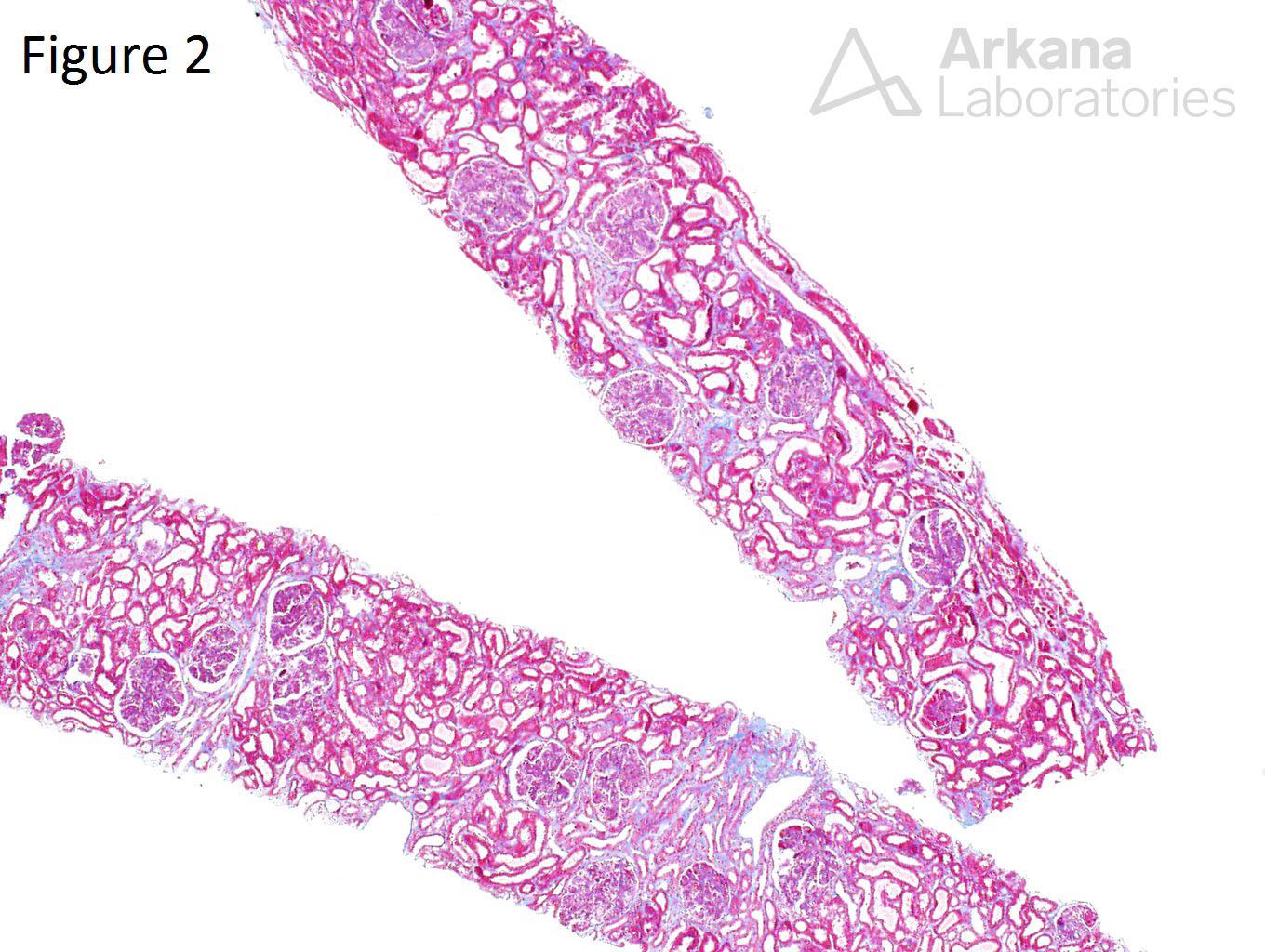
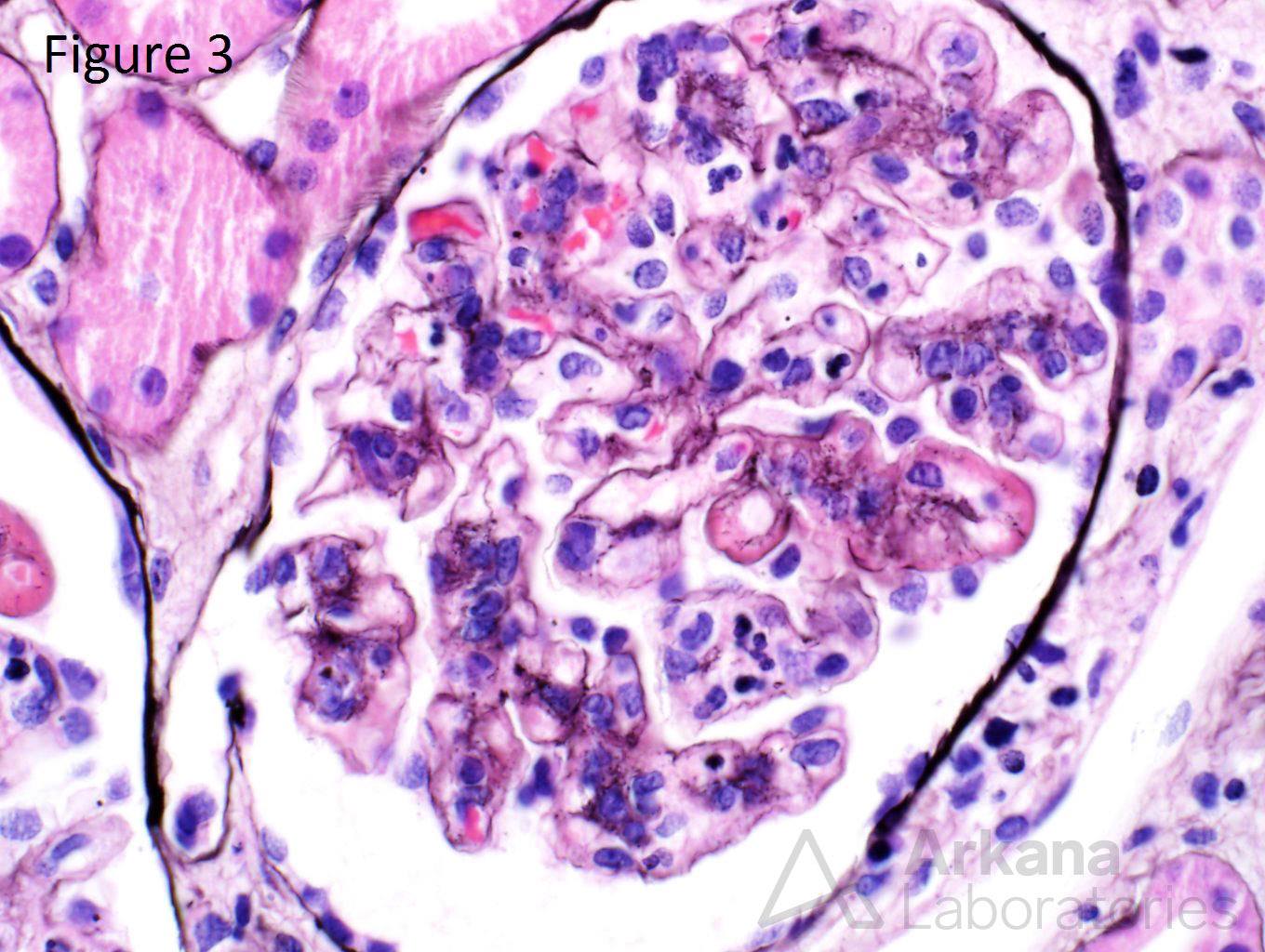
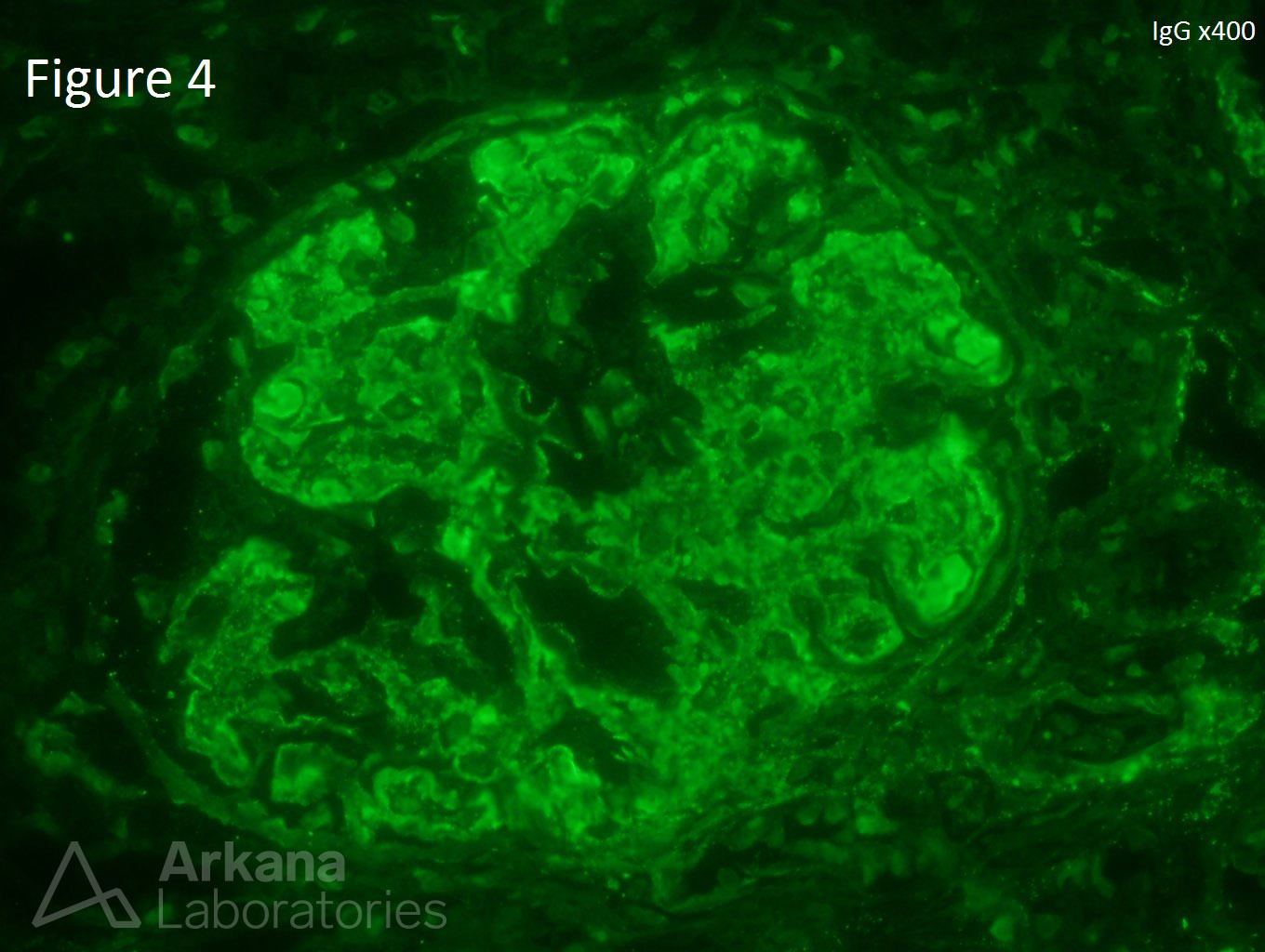
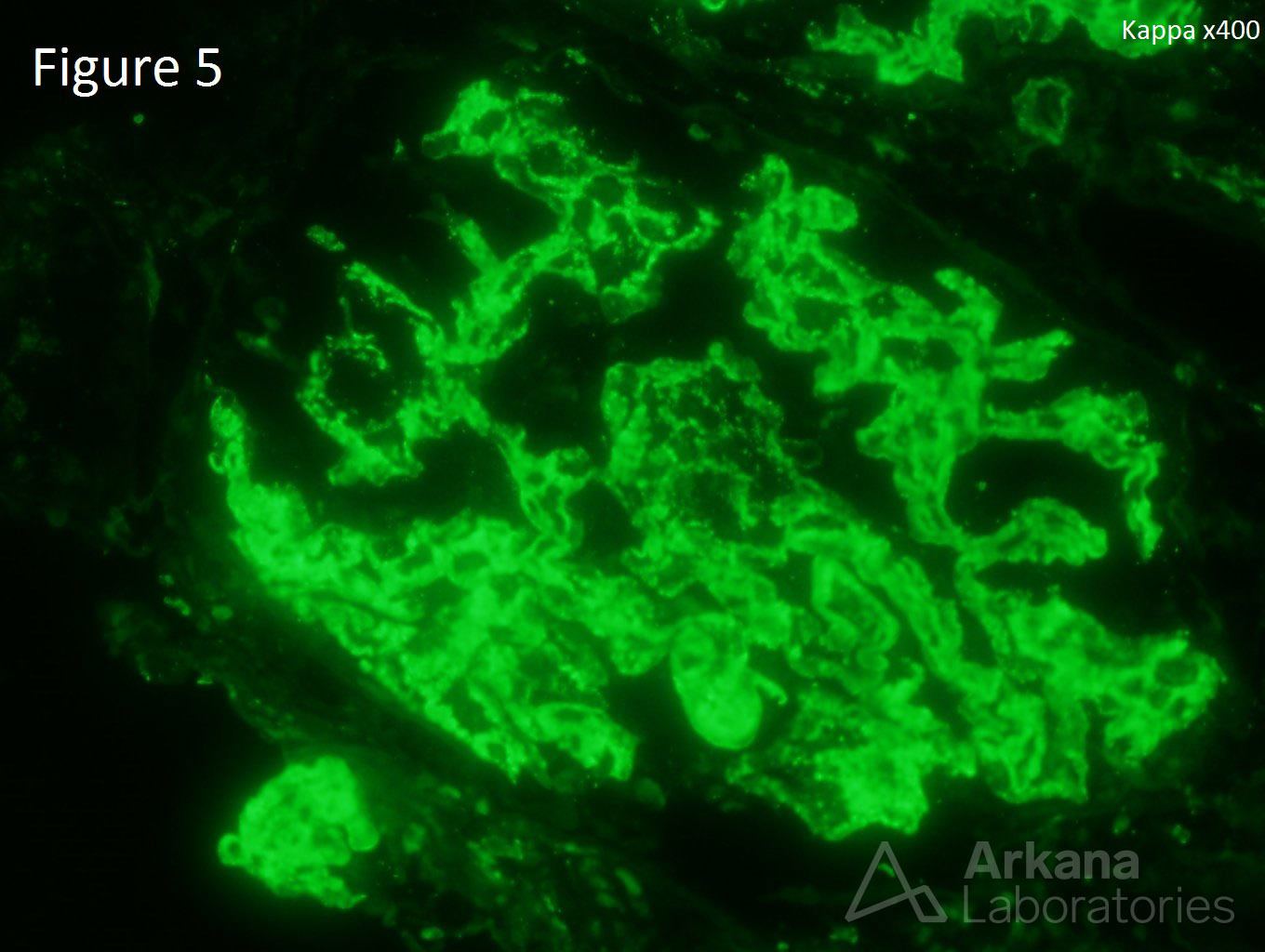
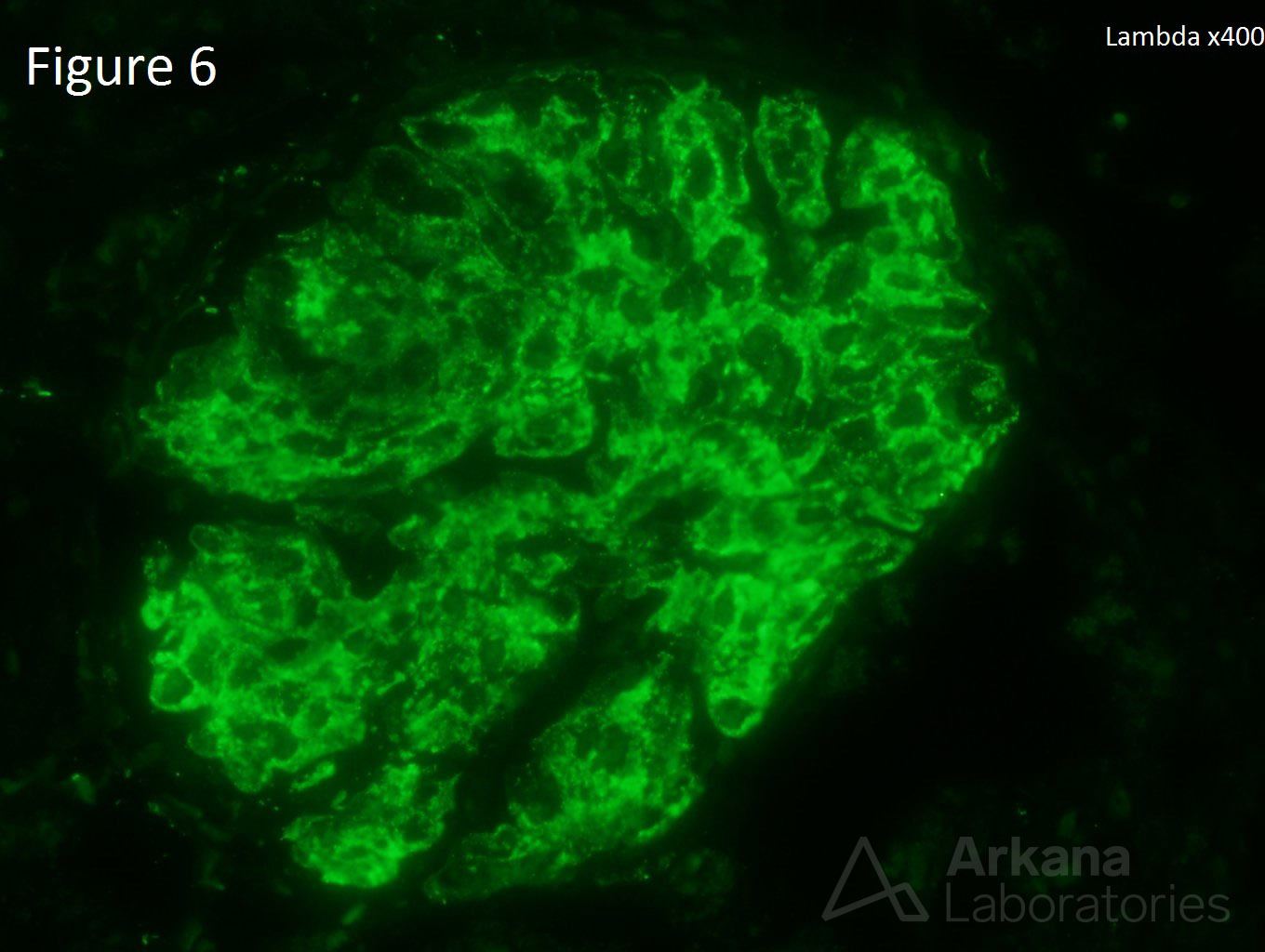
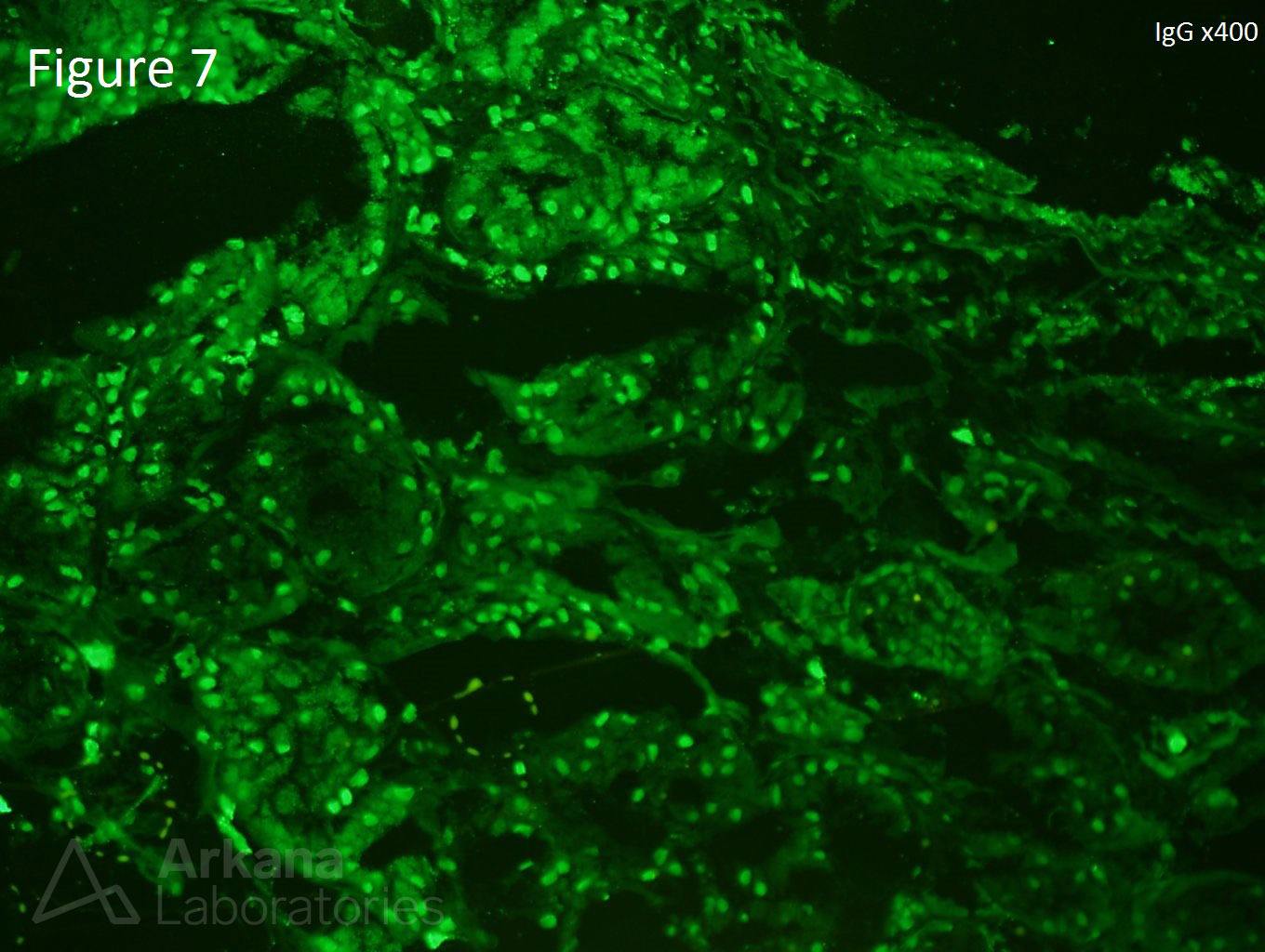
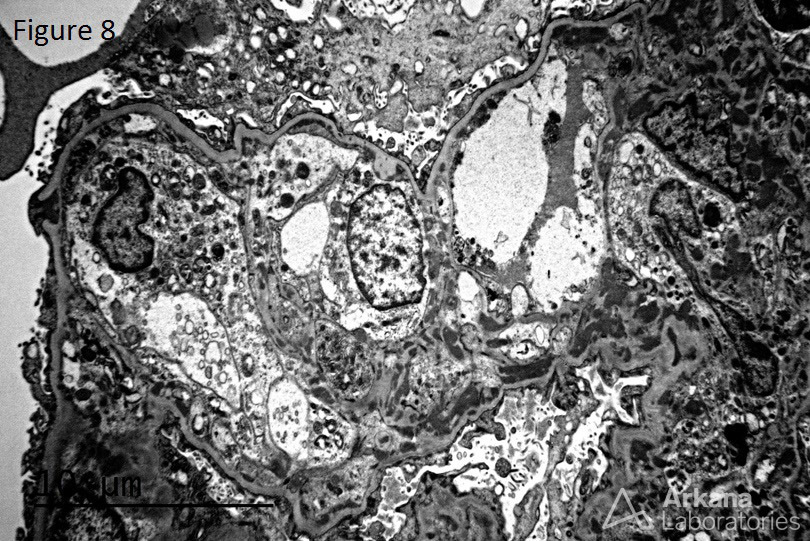
A 20-year-old female presents with hematuria, proteinuria, and a creatinine of 1.2 mg/dl. Serologies for ANA are positive. Complement levels are decreased. She states that auto-immune disease runs in her family, but is unsure about any specific diagnosis. Serologies for dsDNA, SSA, SSB, rheumatoid factor, hepatitis B, hepatitis C, and HIV are pending. Figure 1 shows mesangial and endocapillary hypercellularity with prominent “hyaline thrombi.” Figure 2 shows no significant interstitial fibrosis. Figure 3 shows “wire loops” and no “spikes” and “holes.” Figure 4, Figure 5, and Figure 6 shows mesangial and capillary staining with IgG, kappa, and lambda, respectively. Figure 7 shows a tissue ANA pattern. Figure 8 shows mesangial and endocapillary hypercellularity and immune complex deposits. This is a case of diffuse lupus nephritis (ISN/RPS Class IV).
Lupus is a chronic inflammatory disease that affects the kidneys in 50% of patients. In the United States, there is a predominance of African American and Hispanic patients with lupus nephritis. Unfortunately, worse outcomes including death and ESRD have been found in African American patients when compared to white patients. Altogether, approximately 10% of patients with lupus nephritis develop ESRD. In patients who have class IV lupus nephritis (as described earlier), 44% of patients progress to ESRD within 15 years. Although controversial, protocol repeat kidney biopsies have emerged as a means to inform ongoing treatment decisions and predict long-term renal prognosis. Repeat biopsies have demonstrated considerable discordance between clinically and histologically defined disease activities. Presently, there are no evidence-based guidelines concerning the duration of lupus nephritis maintenance therapy.
Reference:
https://www.ncbi.nlm.nih.gov/pubmed/27821390
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.