NIH Lupus Nephritis Activity and Chronicity Indices: A way to monitor response to treatment and disease progression.
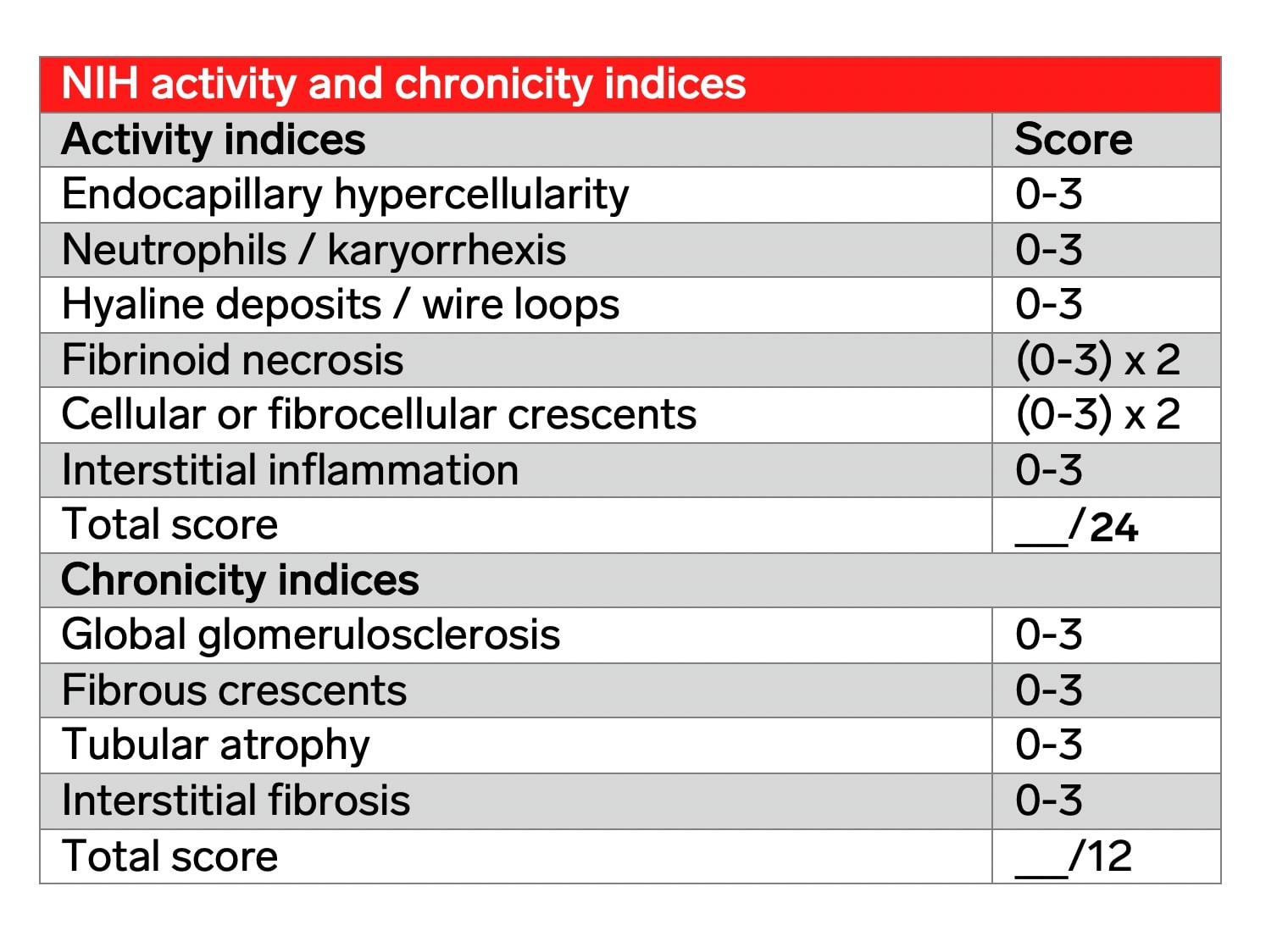
A semi-quantitative grading system of pathologic features on kidney biopsies allows for monitoring response to treatment and showing disease progression. Lupus nephritis disease activity can be assessed on a renal biopsy using the modified NIH activity and chronicity indices. Indicators of disease activity include endocapillary hypercellularity, neutrophils or karyorrhexis within glomerular capillary loops, fibrinoid necrosis, hyaline deposits, cellular or fibrocellular crescents, and interstitial inflammation. Crescents and fibrinoid necrosis are weighted twice as they have a worse impact on prognosis. The scoring is based on the percentage of glomeruli with each feature in the biopsy on a 0 to 3 scale, with a score of 0 = not present, 1 = <25% glomeruli, 2 = 25-50% glomeruli, and 3 indicating >50% glomeruli.
Indicators of disease chronicity include the total percentage of global glomerulosclerosis, fibrous crescents, tubular atrophy, and interstitial fibrosis. Grading the NIH activity and chronicity indices allow comparison in repeat biopsies, as well as for standardized assessment for lupus clinical trials. The criteria for the NIH activity and chronicity indices are shown in the table below and are adapted from Bajema et al, 2018 (reference below). Reporting of these indices is recommended on all kidney biopsies for patients with focal or diffuse lupus nephritis.
References:
https://www.ncbi.nlm.nih.gov/pubmed/29459092
https://ajkdblog.org/2018/06/22/changes-in-isn-rps-classification-of-lupus-nephritis/
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.



