This biopsy came to us for evaluation of nephrotic syndrome. The patient is a Hispanic gentleman in his late 80s, with CKD stage IV and a history of arthritis, diabetes mellitus, coronary artery disease, and hypertension. He had 4.9 g/g of proteinuria. He had negative ANA, hepatitis B, and C serologies. C3 and C4 were within normal limits. Clinically, the differential diagnosis included membranous glomerulopathy and FSGS.
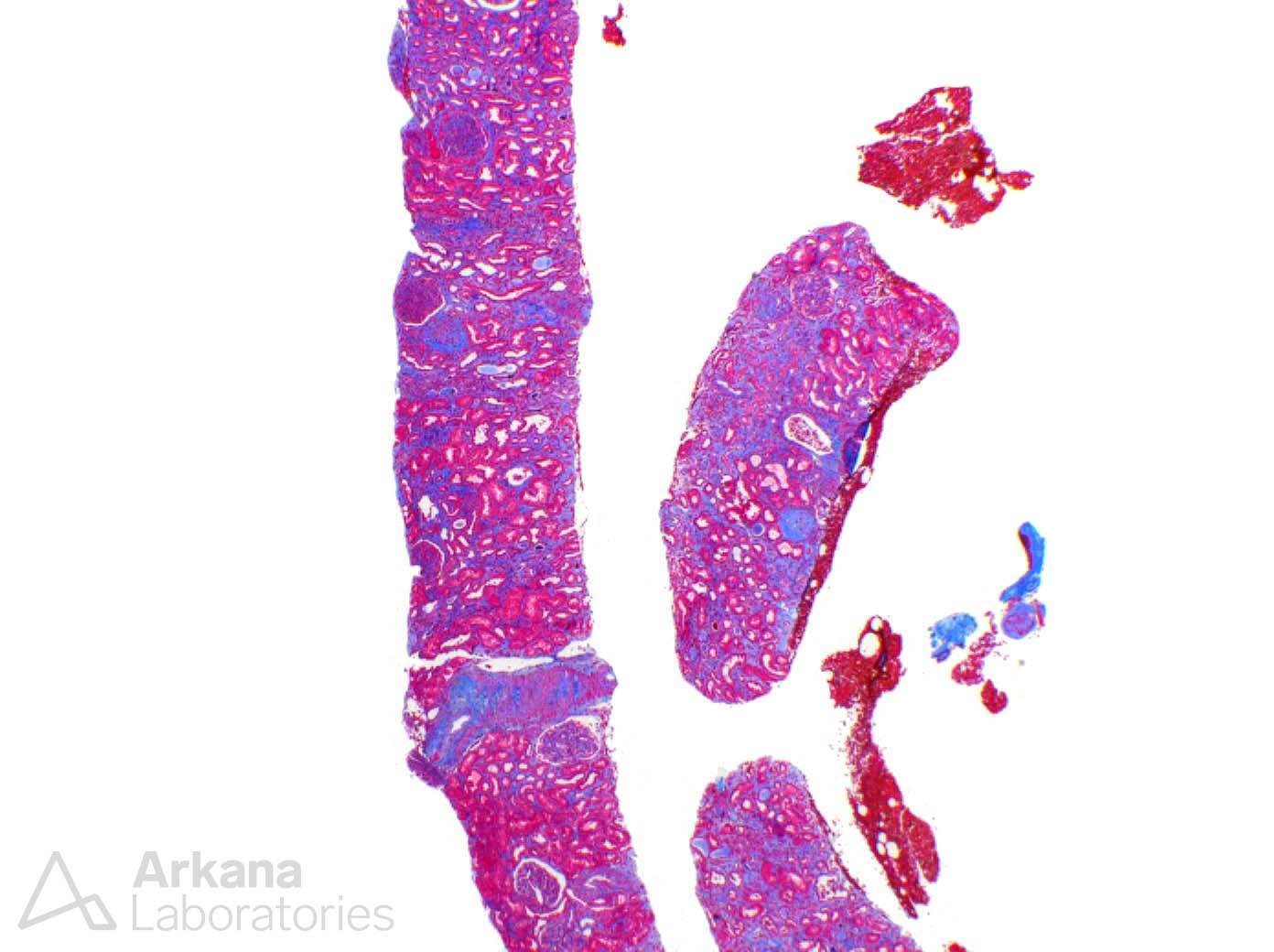
The biopsy was a very good sample, consisting of long cores of renal tissue, mostly from cortex. More than 20 glomeruli were present.
The glomeruli had clear features of diabetic nephropathy, as expected from the patient’s history. Mesangial expansion and nodular glomerulosclerosis were diffusely present. However, lesions of FSGS or the classical features of membranous nephropathy- spikes and holes in the glomerular basement membranes- were not observed.
Patients with progressive diabetic nephropathy may often times have nephrotic range proteinuria, however, true nephrotic syndrome is often times absent. As this patient had a clinical history of nephrotic syndrome, proceeding with a careful biopsy examination is necessary to exclude a second diagnosis occurring on a background of diabetic changes.
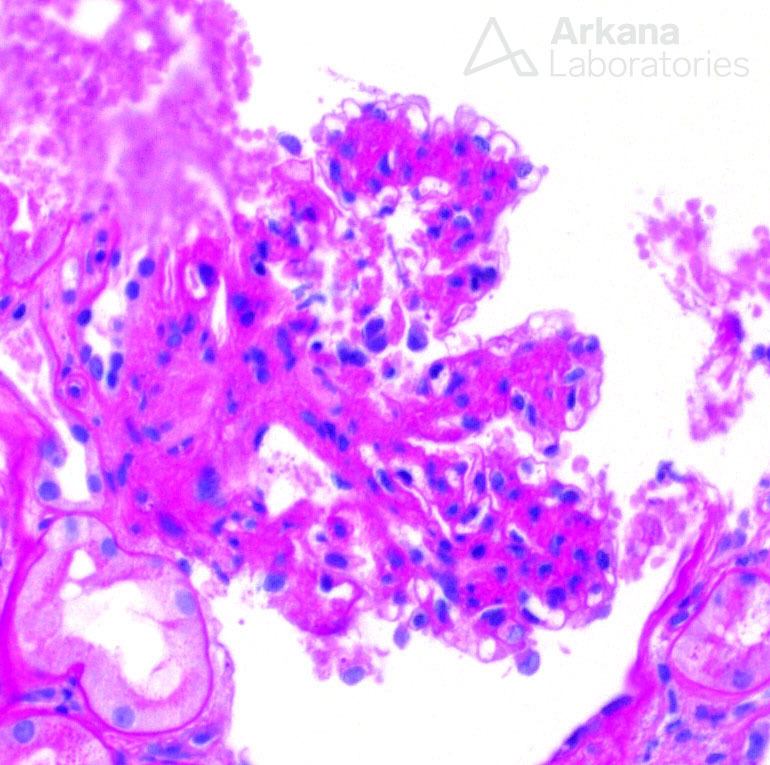
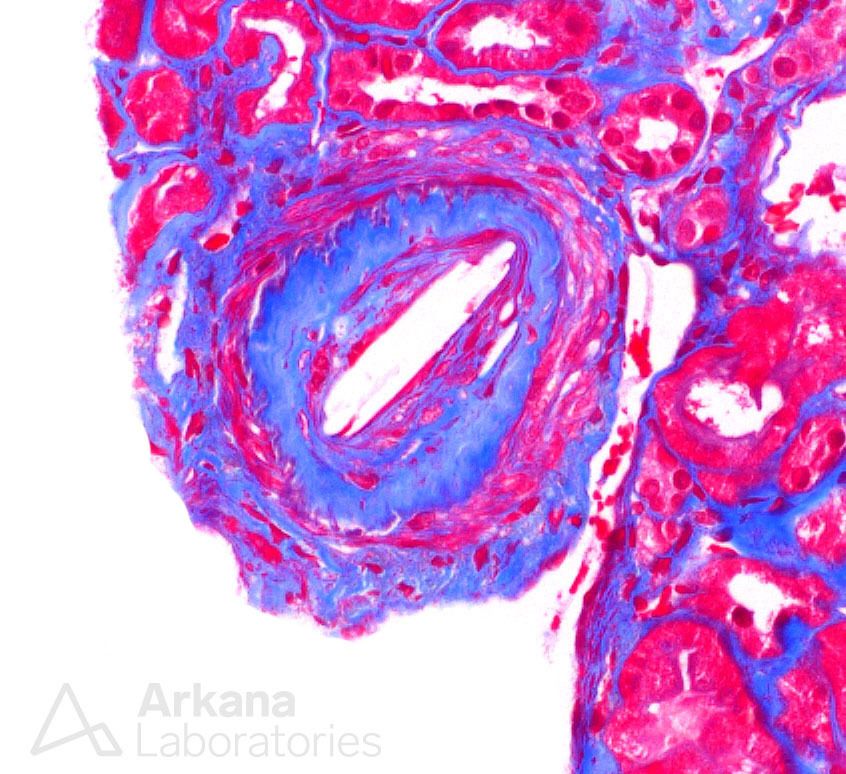
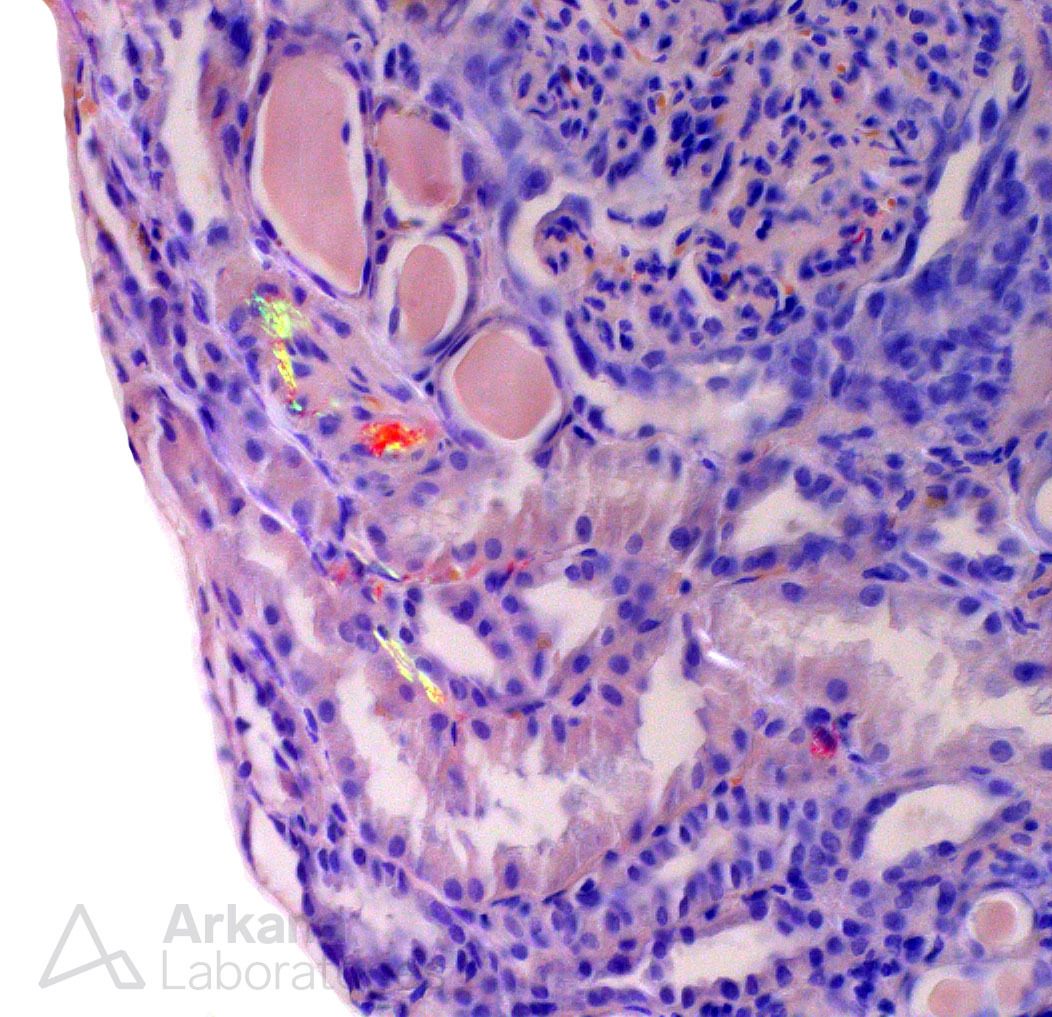
In short order, a second diagnosis was identified within the arteries and arterioles which, while easily seen, was quite important. And in this particular case, the findings were abundant and diffuse throughout the biopsy– cholesterol emboli involving multiple vessels.
At this point, a culprit for the clinical presentation was found, as cholesterol emboli are a potential cause of nephrotic syndrome. The biopsy had not only one but two important diagnoses leading to proteinuria. So, when a third and equally important diagnosis presented itself, one can imagine that it caused surprise.
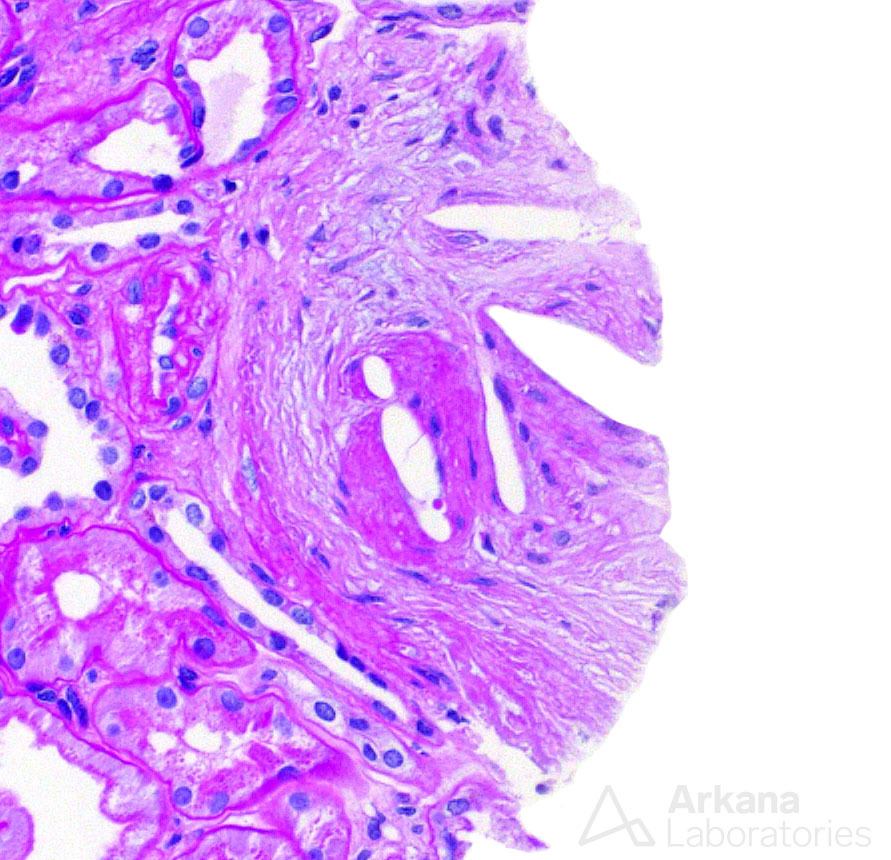
A Congo red stain had been ordered in the biopsy, due to the patient’s age and clinical history. And lo and behold, minute amounts of Congo red positive material were found in arterioles and sparsely distributed in the interstitium. Amyloid!
Due to obvious reasons, a diagnosis of amyloid in a renal biopsy, even to a minimal extent, brings distress to the pathologist and the nephrologist alike. The main cause of amyloidosis in our population is related to plasma cell dyscrasia (AL amyloidosis) and these cases are characterized by light chain restriction on renal biopsy material (more often lambda type).
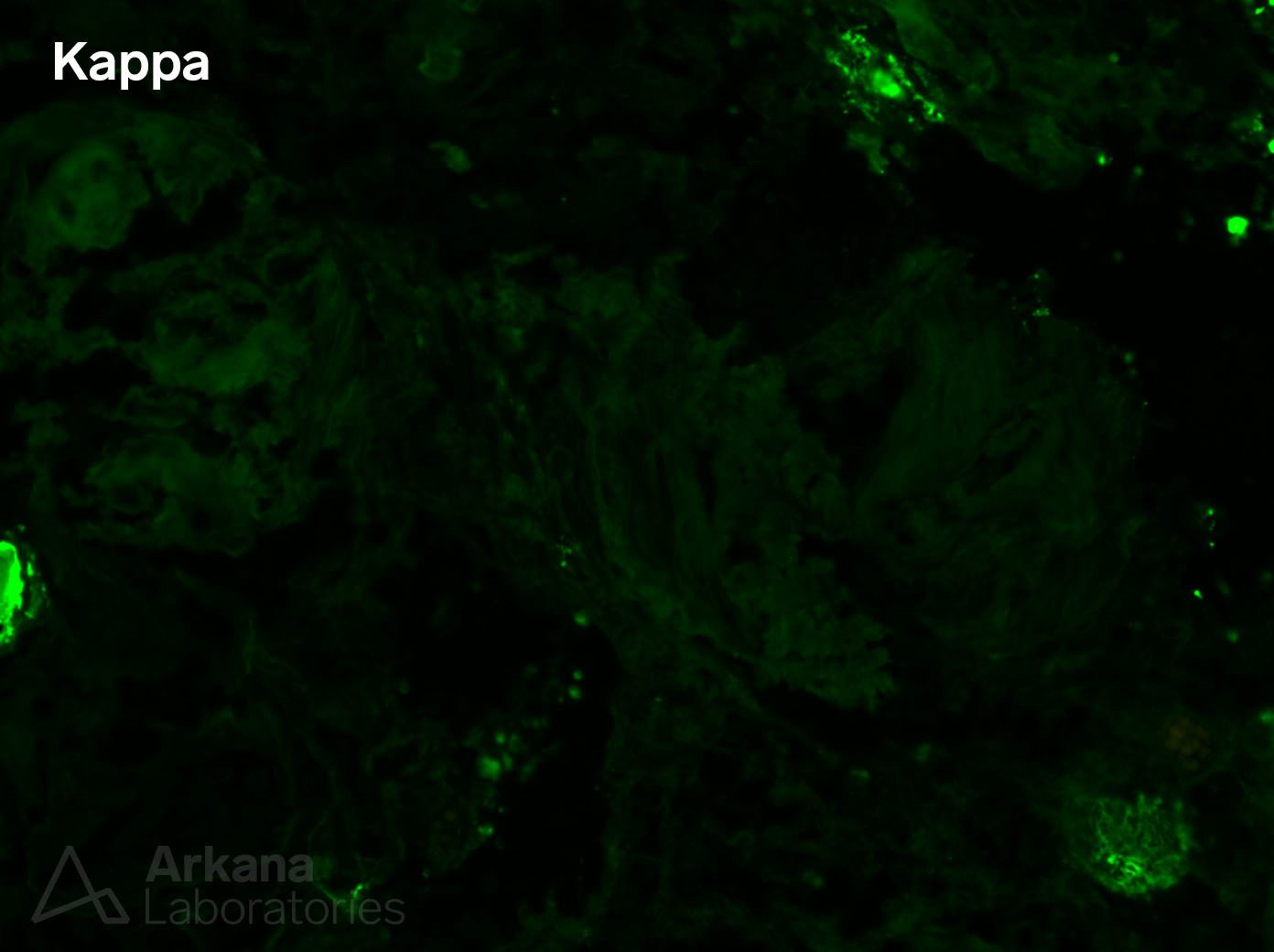
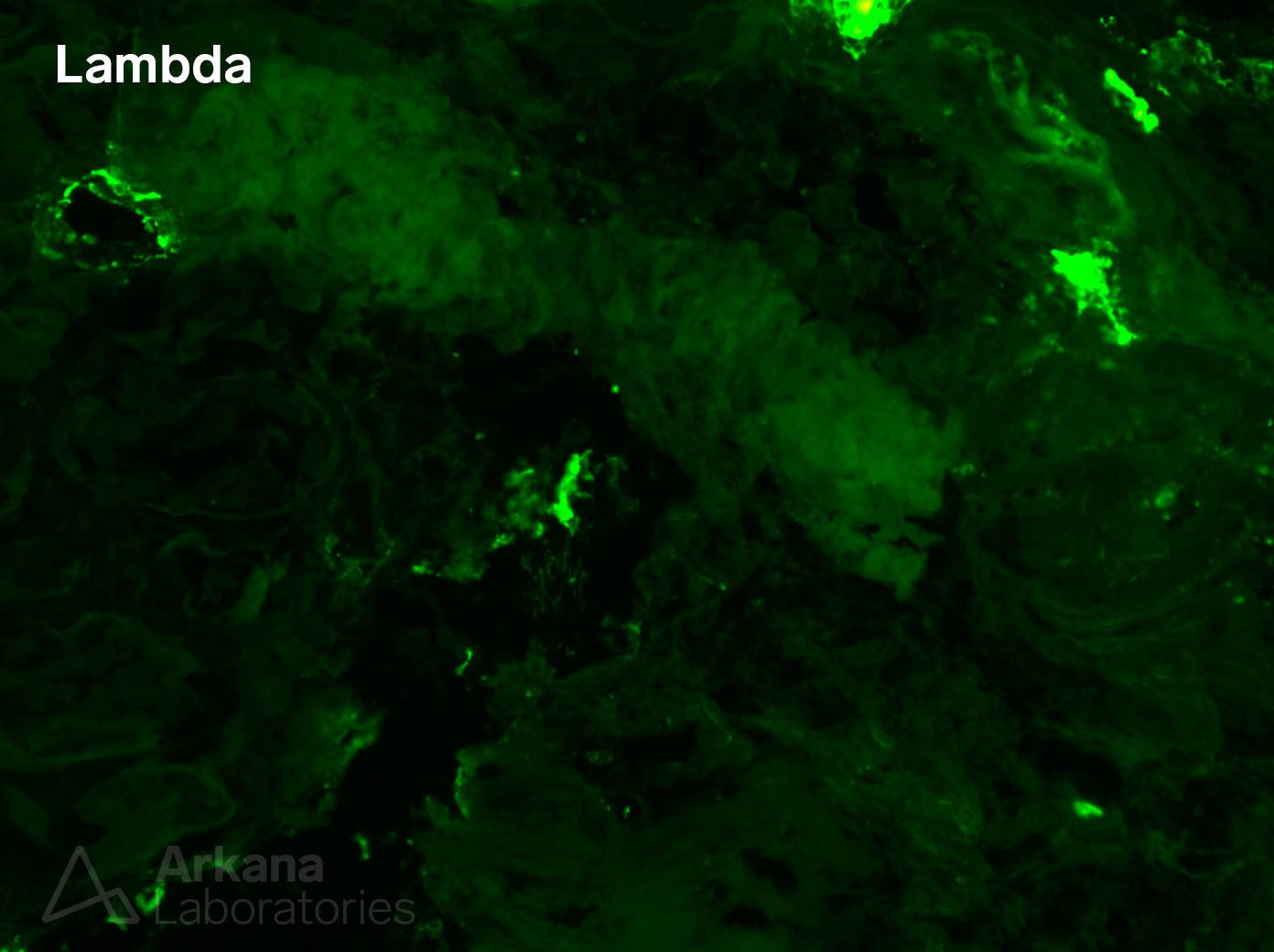
Our immunofluorescence studies, however, showed equal distribution of kappa and lambda light chains throughout the tissue. Furthermore, we did a Congo red stain on the frozen tissue to prove the presence of amyloid deposits on that sample (in small amounts), in the absence of an identifiable light chain restriction.
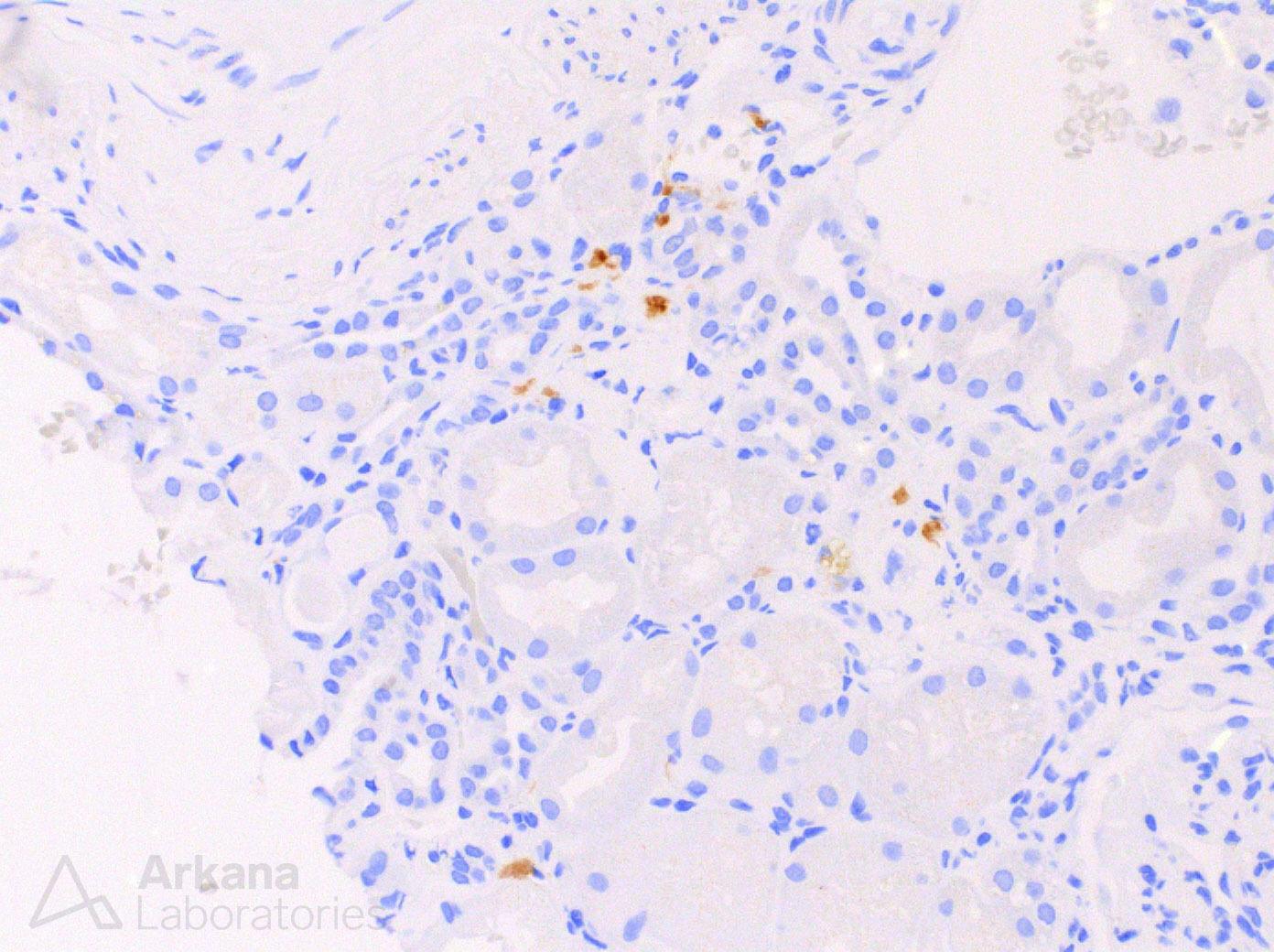
Without light chain restriction, our next steps in typing the amyloid required use of immunoperoxidase stains for other subtypes of amyloid – AA amyloid and LECT2 amyloid.
LECT2 amyloid is not typically characterized by nephrotic syndrome and usually tends to involve the interstitium more than glomeruli. However, LECT2 amyloid is most commonly seen in patients of Hispanic origin, as was the case in this patient. Clinically, it often has an indolent course, and can sometimes be seen in older patients as an incidental finding when biopsied for other issues. Once again, the clinical history and pathology findings work in harmony.
From one to two, to three diagnoses, this renal biopsy was rich in findings. All it required was a bit of digging deeper.
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.