The biopsy is from a 65 year-old female who presented with acute renal failure and elevated creatinine up to 1.9 mg/dl. She has non-ischemic cardiomyopathy and protein electrophoresis is positive for lambda light chains. Serologies for ANA and rheumatoid factor are within normal limits. A biopsy was performed to rule out paraprotein associated disease and there was a specific concern for amyloidosis, due to cardiac changes.
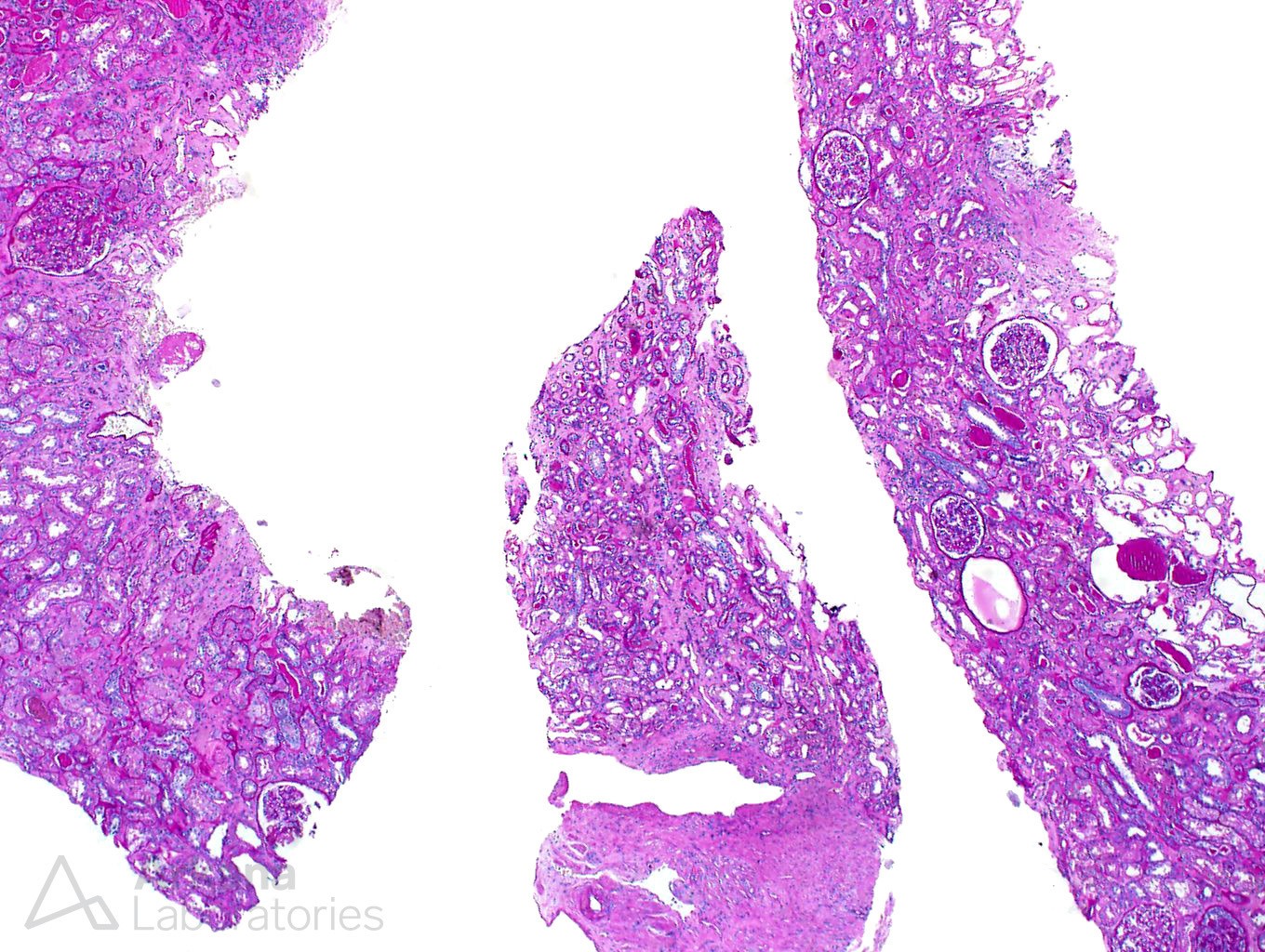
The biopsy consisted of a good sample, contained multiple cores of tissue and more than 20 glomeruli.
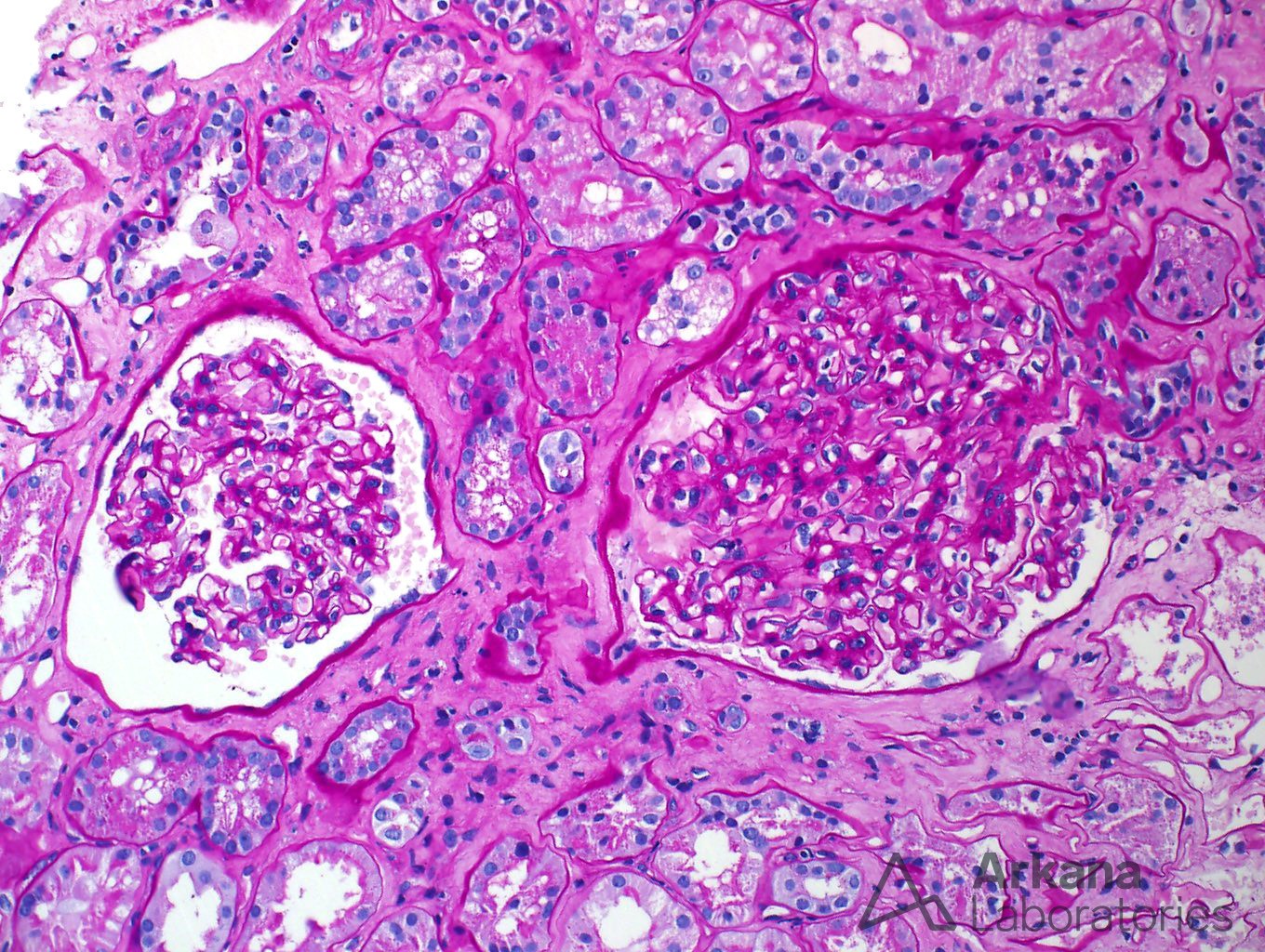
The glomeruli were very unremarkable by light microscopy – loops were open, with regular contours, and no signs of proliferation. Amyloid deposits were not identified in the glomeruli.
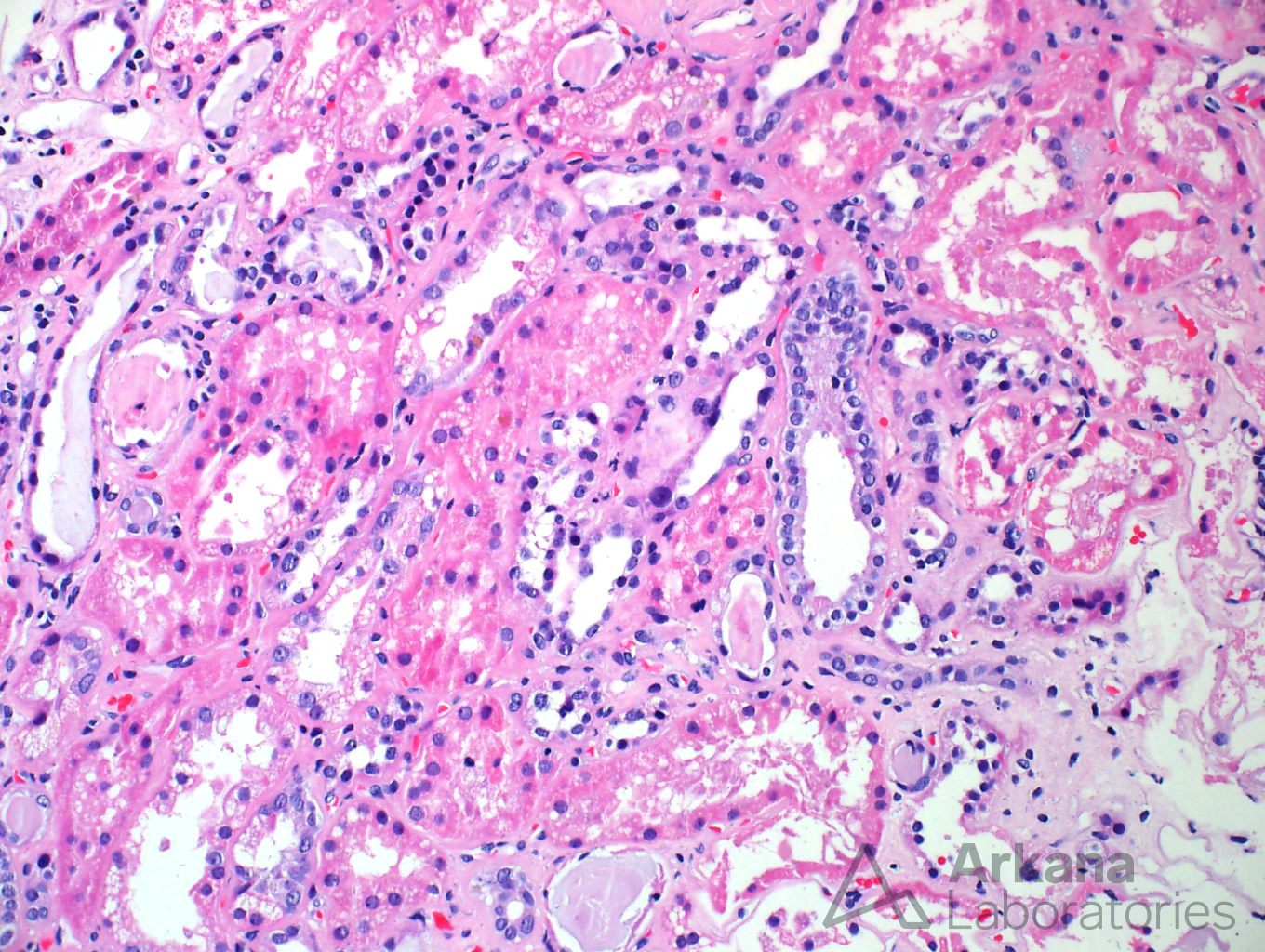
The tubulointerstitium showed signs of tubular injury, with marked cytoplasmic vacuolization, but no other significant changes were noted.
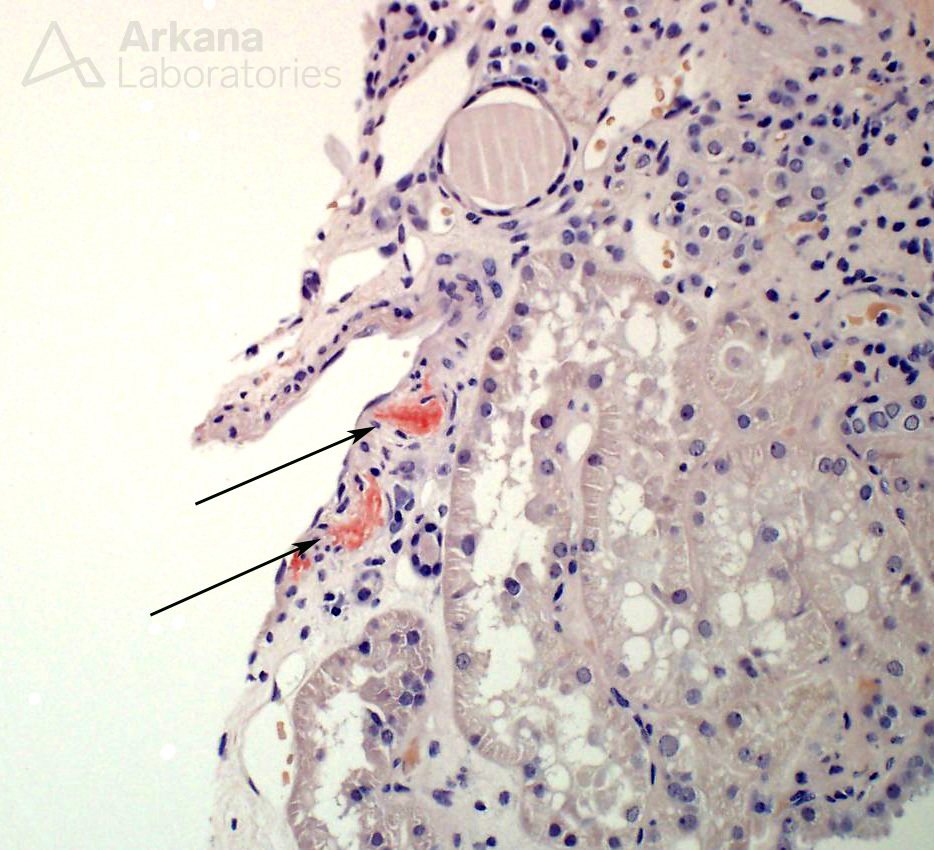
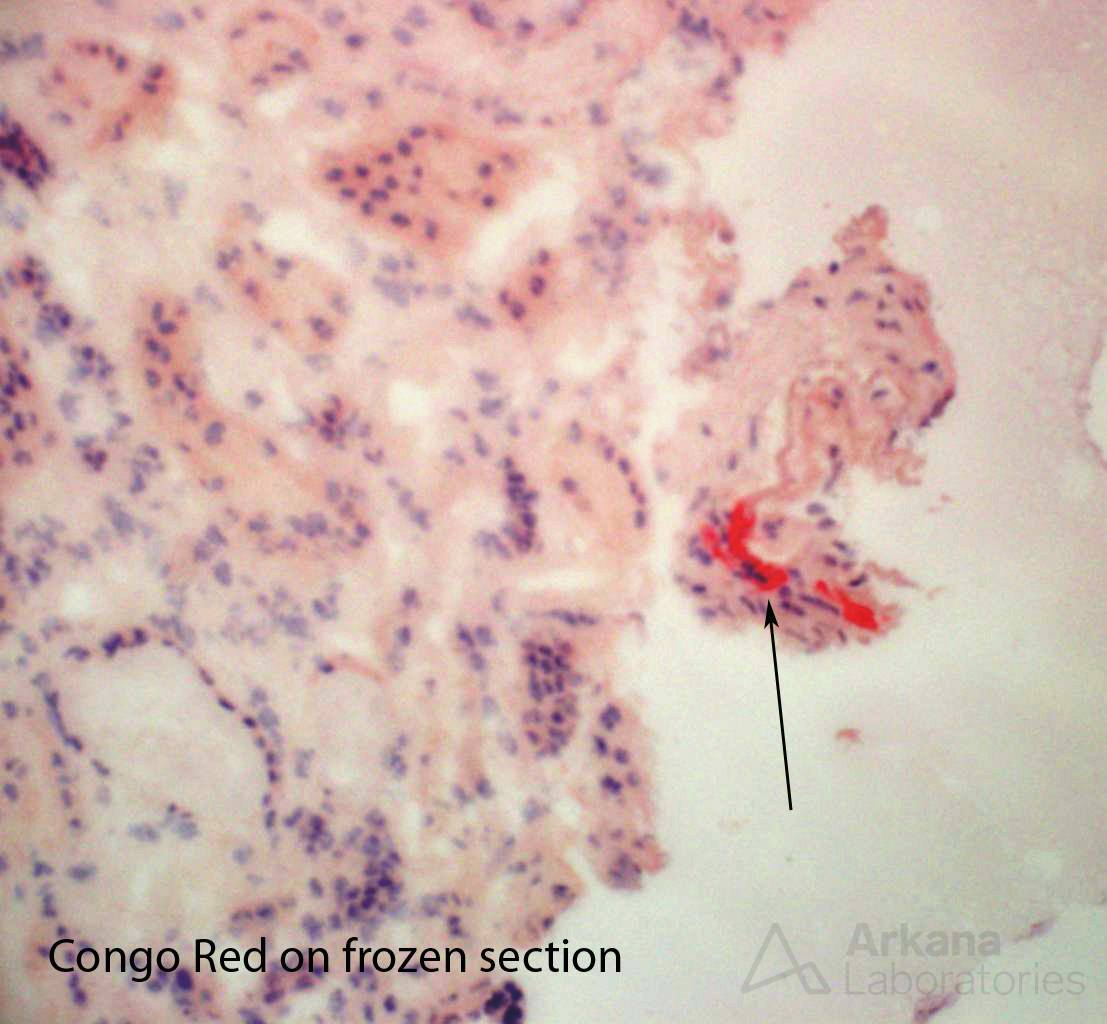
Finally, upon close examination of blood vessels, the clinician’s fears proved justified. In only two small arterioles, there were minimal amounts of amyloid deposits, identified on Congo red stained sections.
The diagnosis of amyloidosis, in this case, was subtle, but not necessarily unexpected. However, a diagnosis of amyloidosis is only partially complete once the amyloid is found. Determining the type of amyloid is as important as finding amyloid on the renal biopsy, if not more so.
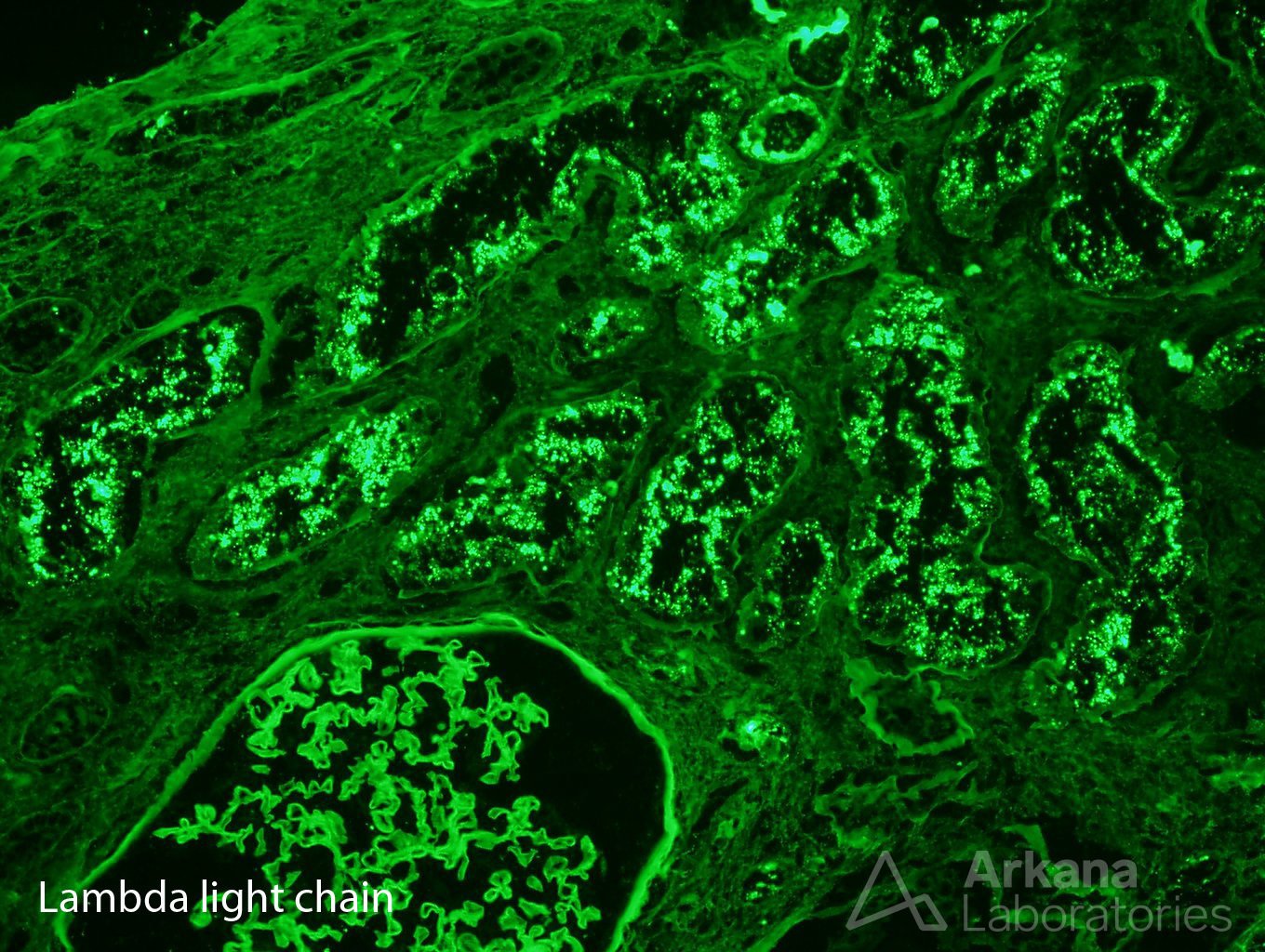
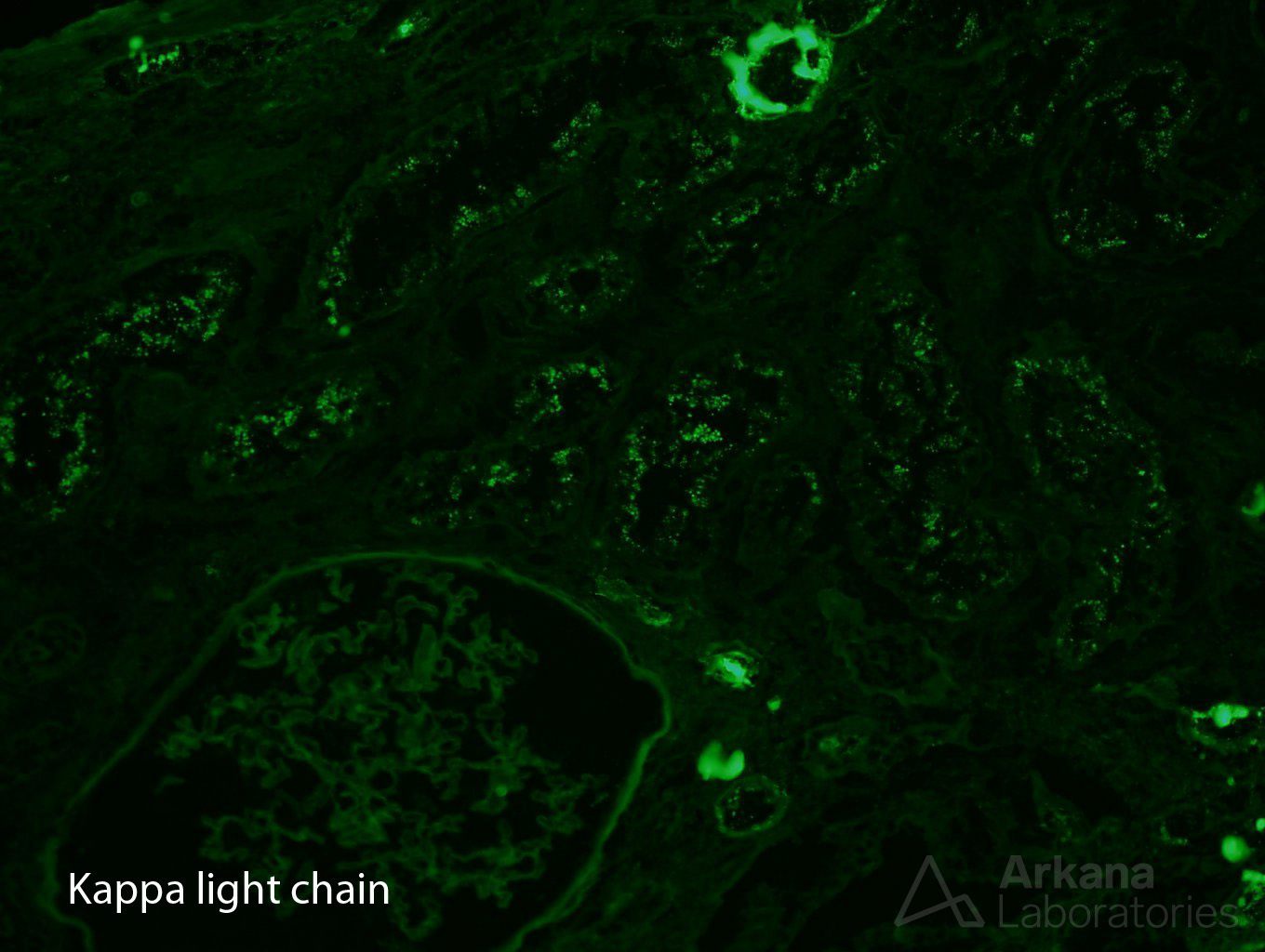
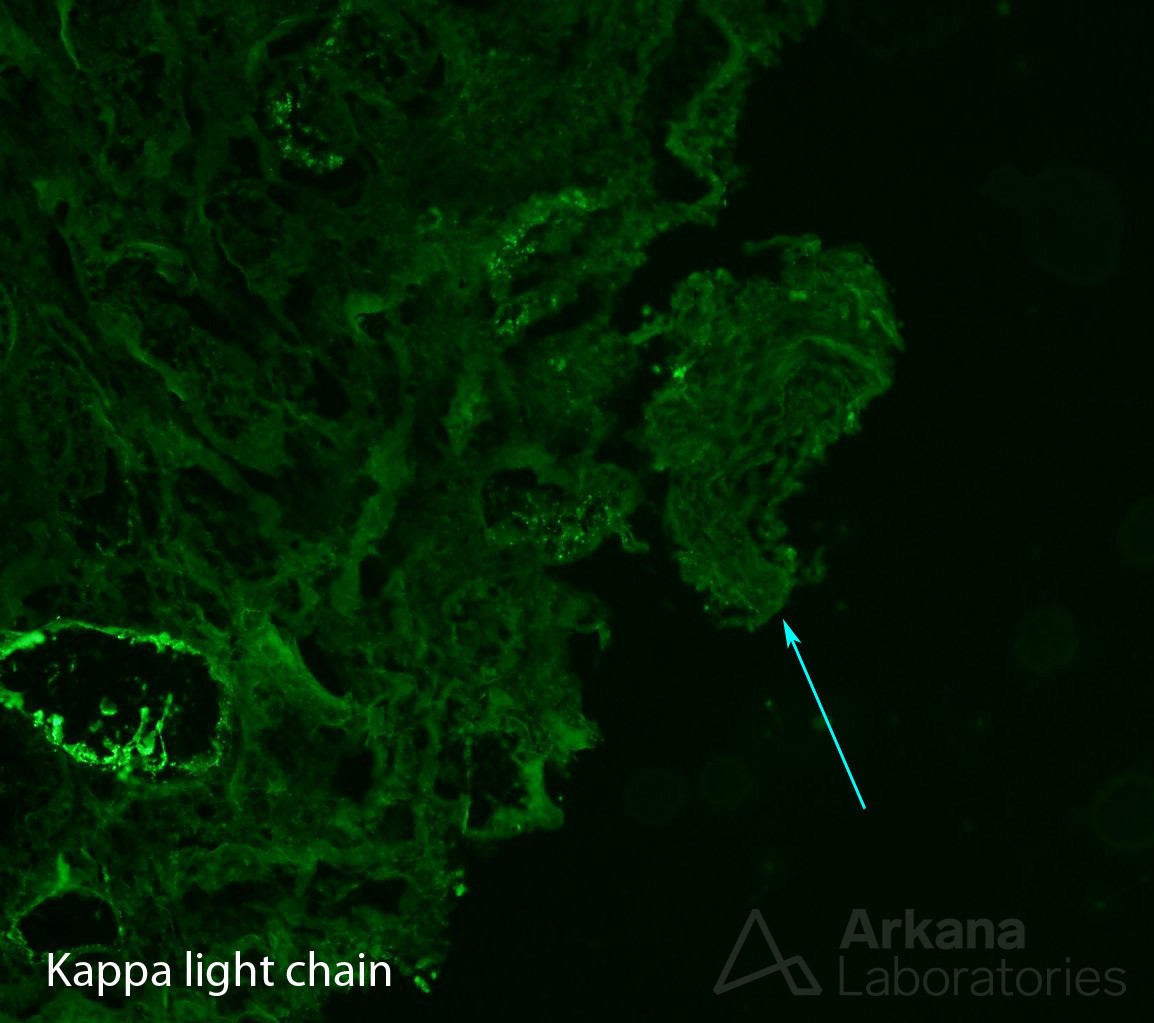
With that in mind, our pathologist looked carefully on immunofluorescence sections, as AL amyloidosis could be proved if kappa or lambda light chains were restricted. And lambda light chain restriction was indeed present, but not as expected. Lambda light chain restriction was limited to protein droplets in proximal tubuli – light chain proximal tubulopathy. And, amyloid deposits could not be seen on darkfield examination.
Now, the pathologist had reached a conundrum in the biopsy – there is amyloid but not definitely proved to be associated to light chain (AL amyloidosis). On the other hand, there is light chain restriction, but not in a pattern of amyloid deposition. What to do?
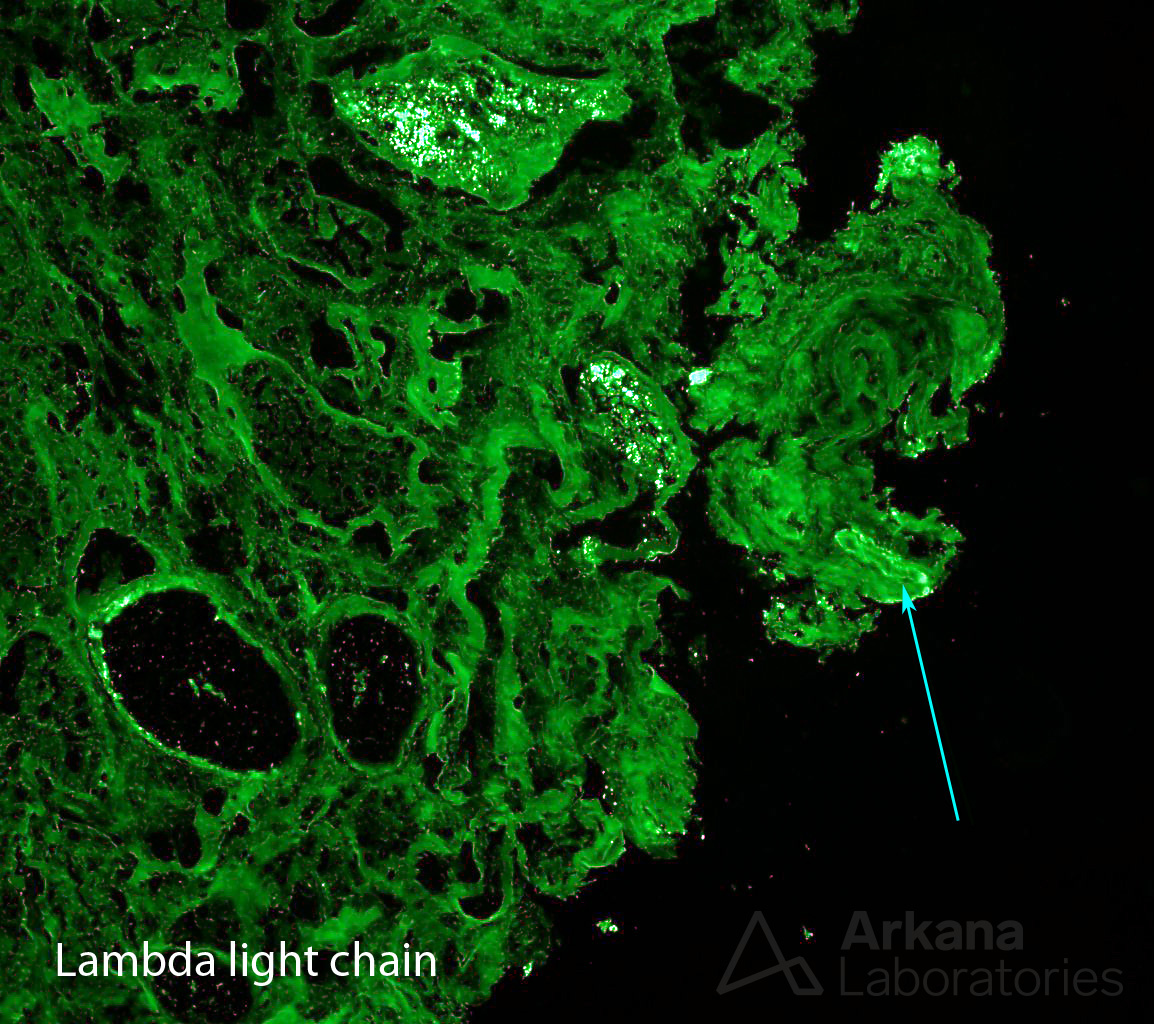
Digging deeper was the answer, but required careful consideration. We needed to identify the presence of amyloid by immunofluorescence, so we could then see if it was light chain restricted. Therefore, we did the unusual step of staining the frozen material (used routinely on immunofluorescence) with a Congo red stain (used routinely on paraffin embedded tissue for light microscopy), along with new sections of kappa and lambda light chains cut in serial after the Congo red. This would allow us to better identify any small deposits of amyloid, and be able to confirm light chain restriction, if present.
Our efforts were worthwhile, and with this triad of stains, a diagnosis of AL amyloidosis, lambda light chain type, was given. Multiple steps and three stains unlocked this dilemma – maybe good things come in threes.
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.