Monday:
Introduction to Systemic Lupus Erythematosus and Clinical Diagnostic Criteria
Answer: C. 50%
50% of adults with SLE will develop lupus nephritis. In children with SLE, 80% have nephritis.
References:
Madhok R. Systemic lupus erythematosus: lupus nephritis. BMJ Clinical Evidence 2015 Dec 18; pp 1123. https://www.ncbi.nlm.nih.gov/pubmed/26683208
Liu CC, Kao AH, Manzi S, Ahearn JM. Biomarkers in systemic lupus erythematosus: challenges and prospects for the future. Therapeutic Advances in Musculoskeletal Disease 2013; 5 (4): 210-233. https://www.ncbi.nlm.nih.gov/pubmed/23904865
In a majority of cases, renal involvement in SLE often occurs in the first year following diagnosis, but can occur at any time in the disease course. #DiseaseWeek
The presence of lupus nephritis increases morbidity and mortality. In a recent retrospective cohort study of 325 patients with systemic lupus erythematosus (SLE) compared to age and gender-matched population controls, SLE patients with nephritis have more than two-fold higher mortality than SLE patients without nephritis. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/31072277
Double-stranded DNA antibody titers help predict lupus nephritis flares if they are present. Double-stranded DNA antibodies are often seen in patients with lupus nephritis and correlate with disease activity. #DiseaseWeek
An increase in dsDNA antibody titer is associated with an increased risk of renal flare and with hypocomplementemia (with an inverse correlation with C3). Antibody titers drop in response to treatment, with a 50% decrease in dsDNA antibody titer reducing renal flare rate 52-53% in two treatment cohorts (Linnik et al, 2005). #DiseaseWeek
Reference: Linnik MD, Hu JZ, Heilbrunn KR, Strand V, Hurley FL, Joh T, LJP 394 Investigator Consortium. Relationship between anti-double-stranded DNA antibodies and exacerbation of renal flares in patients with Systemic Lupus Erythematosus. Arthritis Rheum 2005 April; 52 (4): 1129-1137.
https://www.ncbi.nlm.nih.gov/pubmed/15818711
Anti-Smith antibodies are also highly specific for SLE, but have poor sensitivity (29%). Serum anti-Smith antibodies correlate with membranous lupus nephritis. #DiseaseWeek

Reference: Field M, Williams DG, Charles P, Maini RN. Specificity of anti-Sm antibodies by ELISA for systemic lupus erythematosus: increased sensitivity of detection using purified peptide antigens. Ann Rheum Dis 1988 Oct; 47 (10): 820-825. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1003610/
Lupus pathogenesis is caused by dysfunction in T-cell and B-cell activation. Lupus T cells show mitochondrial dysfunction and reduced generation of ATP, which predisposes to necrotic versus apoptotic cell death. #DiseaseWeek
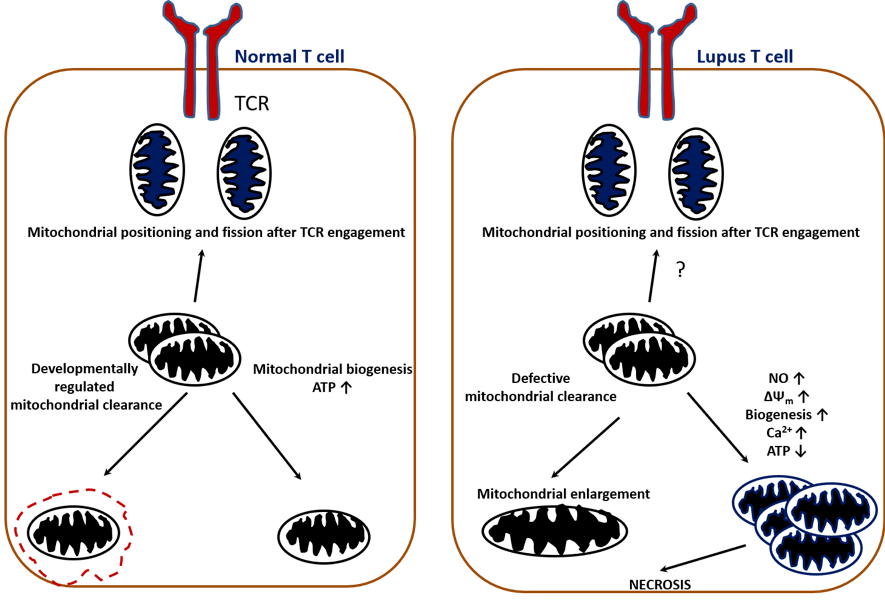
Reference: Caza TN, Talaber G, Perl A. Metabolic regulation of organelle homeostasis in lupus T cells. Clinical Immunology 2012 Jul 13, 144 (3): 200-213. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3423541/
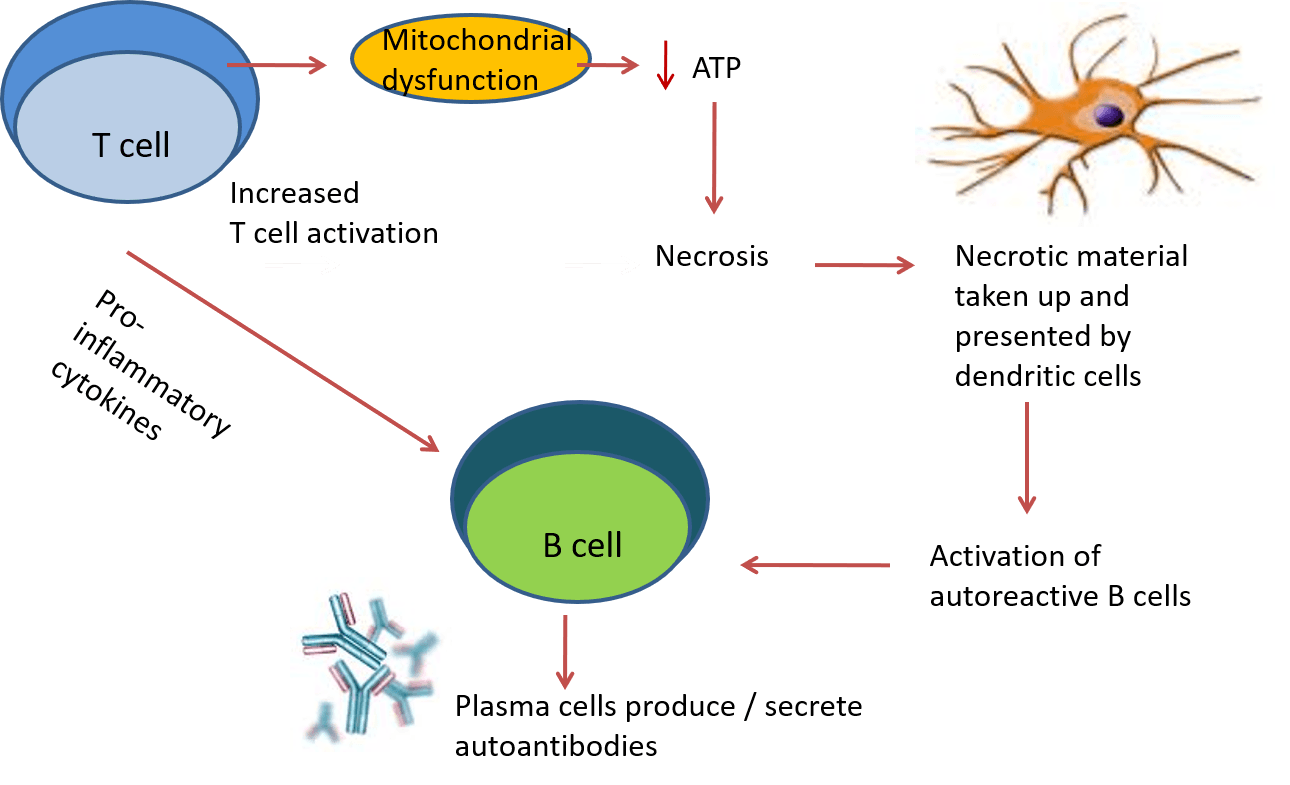
Antigen-presenting cells take up necrotic material and become activated through toll-like receptors. This leads to activation of autoreactive B cells, which differentiate into plasma cells and secrete autoantibodies (see image below). #DiseaseWeek
Infections may trigger autoimmunity in SLE. Molecular mimicry of microbial components with self-antigens can result in the activation of autoreactive T cells. This can result in cytokine and antibody production against self-antigens. #DiseaseWeek
Epitope spreading (both intramolecular and intermolecular) results in amplification of this inflammation. Additionally, superantigens (bacterial, viral) can non-specifically activate T lymphocytes, which further promotes inflammatory responses. #DiseaseWeek
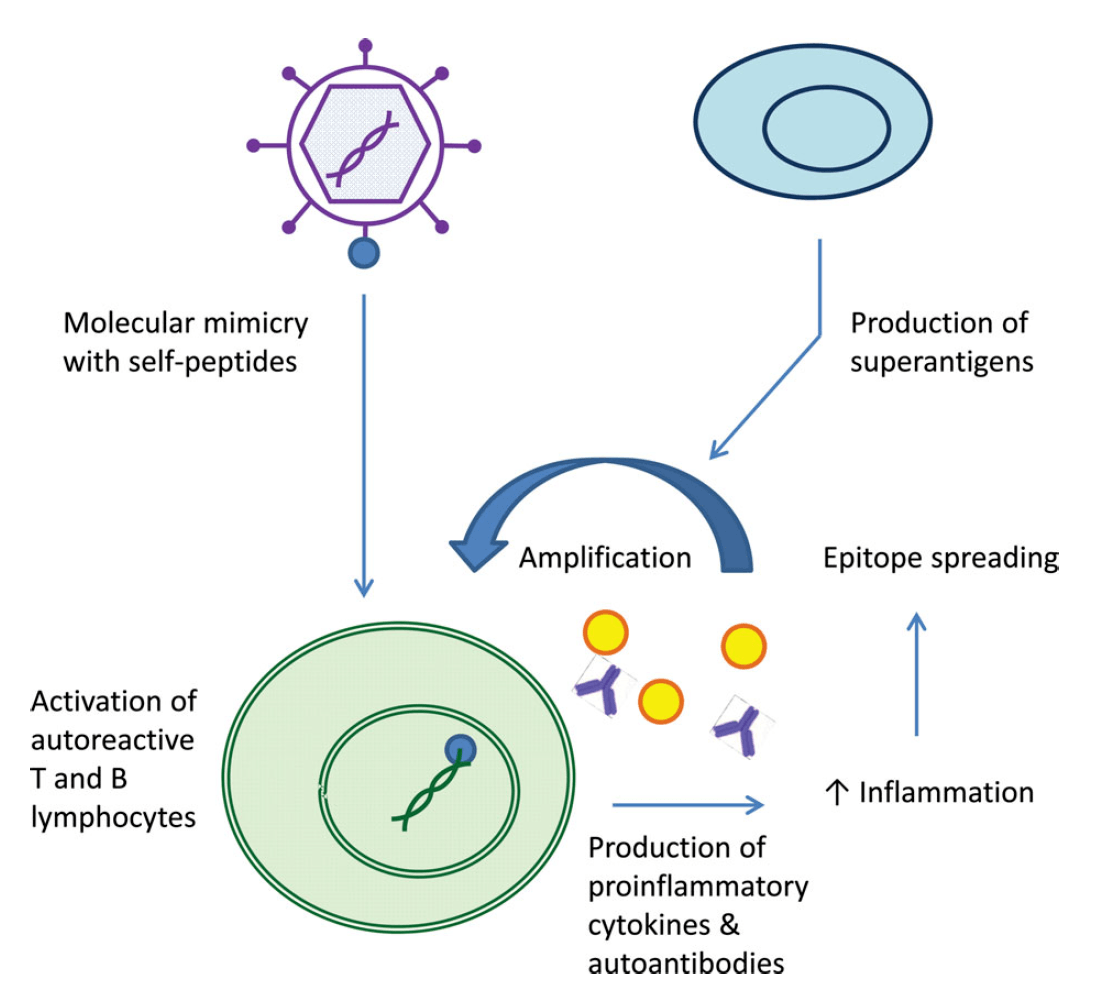
Reference: Caza T. Oaks Z, Perl A. Interplay of infections, autoimmunity, and immunosuppression in Systemic Lupus Erythematosus. International Reviews of Immunology 2014; 1-34. https://www.ncbi.nlm.nih.gov/pubmed/24471448
Tuesday:
International Society of Nephrology / Renal Pathology Society Classification of Lupus Nephritis
Answer: D. ISN/RPS class III + V
Concurrent membranous lupus nephritis (ISN/RPS class V) can be diagnosed with proliferative lupus nephritis (class III or IV) if >50% of the glomerular capillary loops in >50% of glomeruli show subepithelial and/or intramembranous IgG deposits.
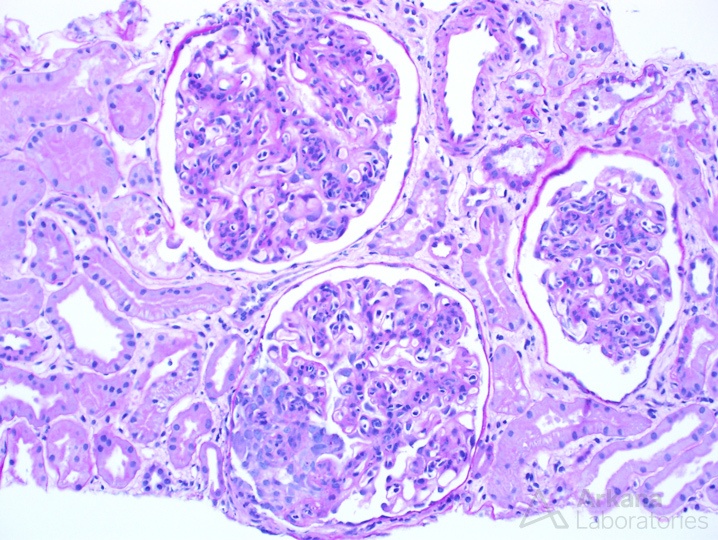
The photomicrograph above shows three glomeruli, all with endocapillary proliferation and one showing a nascent cellular crescent. These findings are seen in proliferative lupus nephritis. #DiseaseWeek
Proliferative lupus nephritis, either focal (ISN/RPS class III) or diffuse (ISN/RPS class IV) is determined by the presence immune deposition along with of active and chronic lesions on kidney biopsies. There is typically “full house” immunofluorescence, with the presence of all three immunoglobulin heavy chains (IgA, IgM, and IgG) and complement (C3 and C1q). #DiseaseWeek
Active lesions of lupus nephritis include endocapillary hypercellularity, karyorrhexis / intracapillary neutrophils, fibrinoid necrosis, rupture of the glomerular basement membrane, crescents (cellular or fibrocellular), presence of subendothelial deposits (wire loops), and intraluminal hyaline thrombi / immune aggregates. #DiseaseWeek
Chronic lesions of lupus nephritis include global and segmental glomerulosclerosis, fibrous crescents, and fibrous adhesions. At least one glomerulus needs at least one of the active lesions for a diagnosis of focal lupus nephritis. For diffuse lupus nephritis, greater than 50% of glomeruli are involved by any of these features (single or in combination). #DiseaseWeek
In the previous lupus nephritis classification, segmental and global glomerular lesions were separated out. These are now no longer a component of the classification, but instead it is recommended to provide the NIH activity and chronicity indices. #DiseaseWeek
The presence of “full house” immunofluorescence with proliferative lesions can occur with lupus nephritis, but also can be seen in infections, including HIV (HIV-associated immune complex disease of the kidney). In the absence of other clinical features of SLE, an infectious etiology should be ruled out. #DiseaseWeek
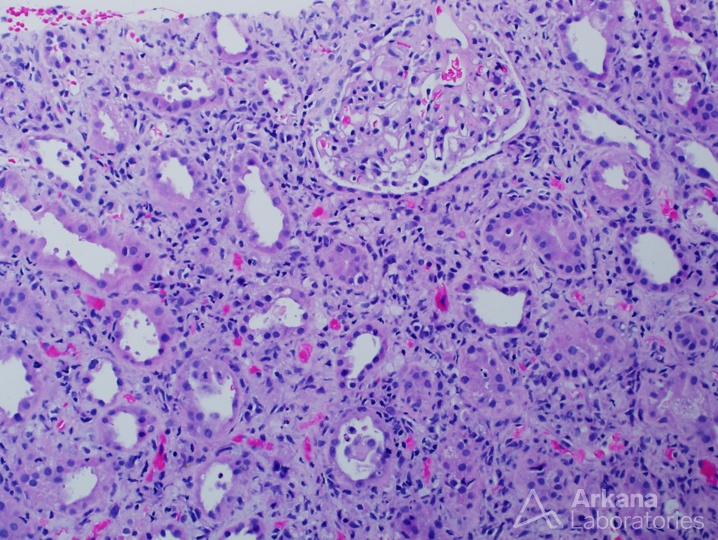
Acute interstitial nephritis with concurrent mesangial proliferative lupus nephritis (ISN/RPS class II)
Concurrent tubulointerstitial disease can occur in any type of lupus nephritis, but is most common in class III and IV disease. Tubulointerstitial disease includes interstitial inflammation, edema, and mixed infiltrates. Chronic tubulointerstitial disease results in IFTA. #DiseaseWeek
Immune deposits can occur along tubular basement membranes. There is no correlation between the degree of deposition and inflammation/scarring. https://www.ncbi.nlm.nih.gov/pubmed/3540691
Answer: B. Focal lupus nephritis. Wire loops due to subendothelial immune deposits, and hyaline thrombi (detached subendothelial deposits in a capillary loop) are seen in focal and diffuse lupus nephritis (class III and IV), and not within minimal mesangial, mesangial proliferative, or membranous lupus nephritis. A “wire loop” is shown below by electron microscopy:
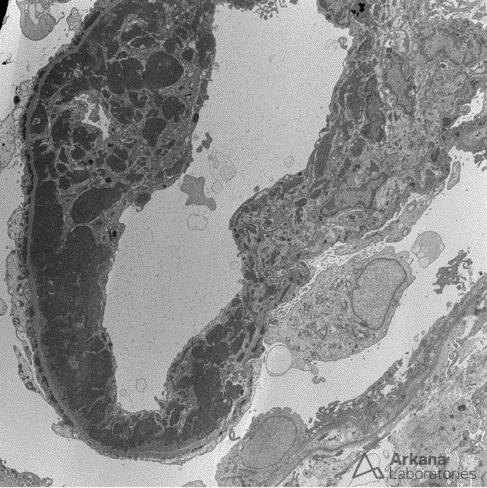
Large confluent subendothelial deposits result in the appearance of “wire loops” by light microscopy.
Tubuloreticular inclusions are subcellular structures seen within the endoplasmic reticulum at the subendothelial surface that are frequently associated with lupus nephritis.
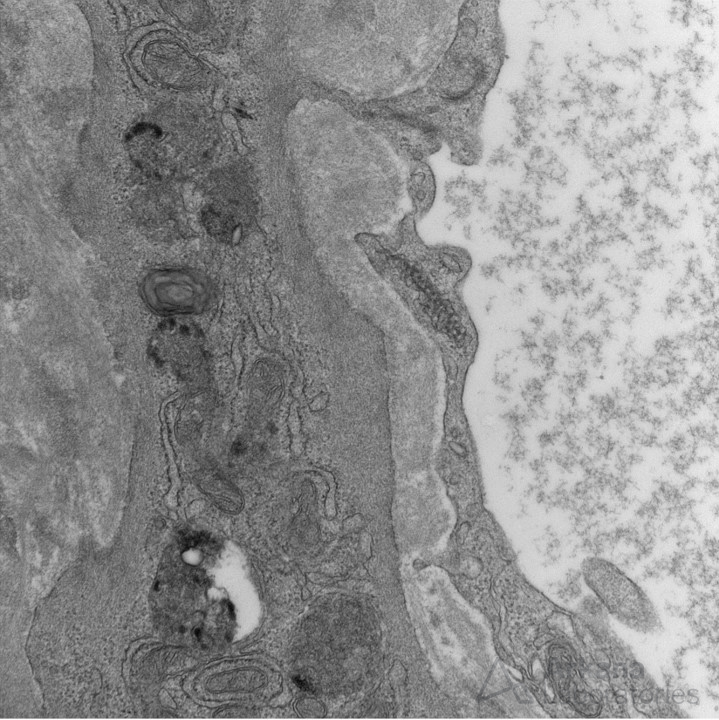
The formation of tubuloreticular inclusions is attributed to exposure of excess type I interferon (endogenous, exogenous, or via interferon therapy (such as for viral hepatitis). While these structures are associated with SLE, they are not specific and have poor sensitivity.
In a kidney biopsy series of 104 cases with tubuloreticular inclusions, the authors found that 59.6% had lupus nephritis, 19.2% had a viral etiology, and the remaining had IgA nephropathy, HSP, ANCA, and rare cases of diabetic nephropathy. https://www.ncbi.nlm.nih.gov/pubmed/23957425
Cytotoxic agents, including intravenous cyclophosphamide has been shown to be superior to high dose steroids for the treatment of proliferative lupus nephritis and decreases the risk of end-stage kidney disease (Austin et al, 1986). #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/3511372
IV methylprednisolone + IV cyclophosphamide increases tx efficacy. 29% of lupus nephritis pts achieved remission with IV methylprednisolone, and 62% with cyclophosphamide, but 85% remitted using combination therapy (Gourley et al, 1996). #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/8815753
There were superior outcomes with induction, followed by maintenance therapy. Induction therapy includes either cyclophosphamide or mycophenolate mofetil, while maintenance therapy is with azathioprine and prednisone. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/8815753
Mycophenolate mofetil (MMF) and methylprednisolone were found to be as effective as IV cyclophosphamide and prednisolone. African American and Hispanic patients had better outcomes with MMF compared to Caucasian patients. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/19369404
The first biological therapy for lupus nephritis is Belimumab, a monoclonal ab that inhibits the B-cell stimulator BAFF. When added to standard therapy, belimumab reduced dz activity, reduced severe flares, and was well tolerated. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/21296403
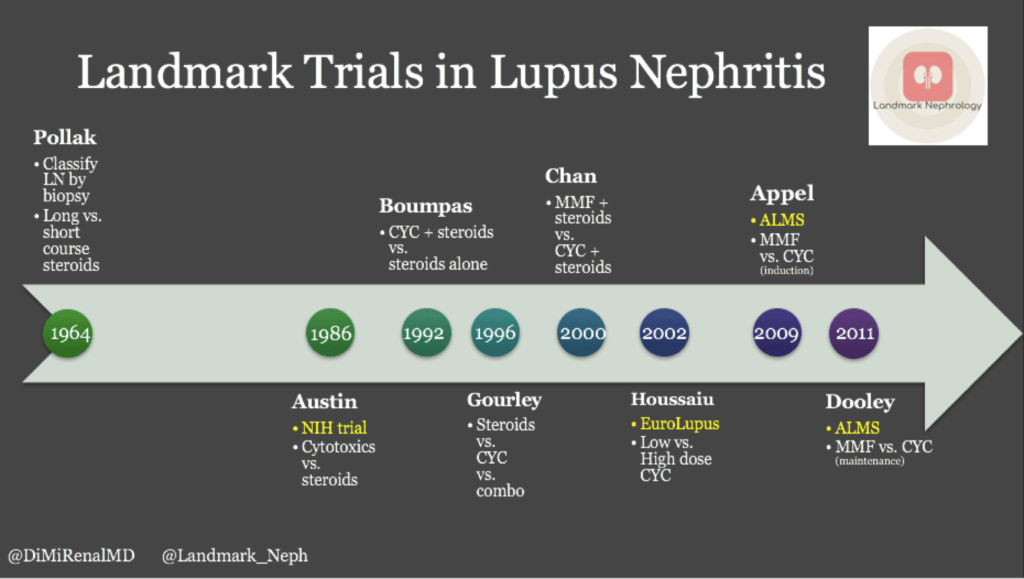
A nice timeline of trials in treatment of lupus nephritis can be found on @RenalFellowNtwk by @DiMiRenalMD at https://www.renalfellow.org/2019/01/09/landmark-trials-in-lupus-nephritis-look-how-far-weve-come/
https://www.ncbi.nlm.nih.gov/pubmed/3511372
https://www.ncbi.nlm.nih.gov/pubmed/8815753
https://www.ncbi.nlm.nih.gov/pubmed/19369404
https://www.ncbi.nlm.nih.gov/pubmed/21296403
Wednesday:
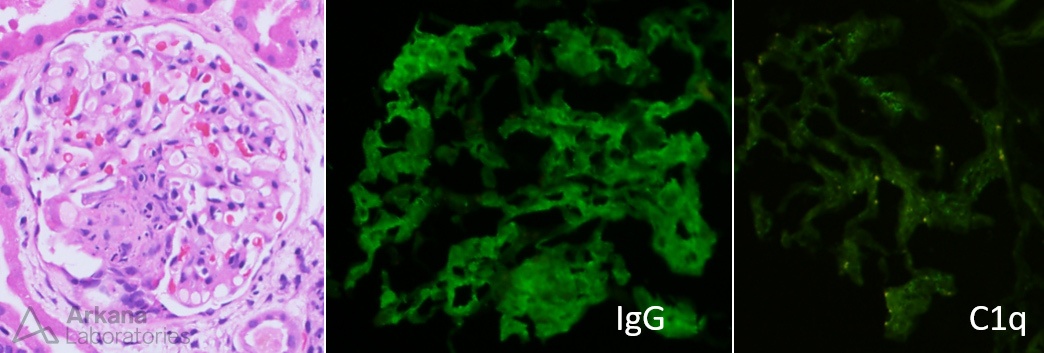
The image shows granular mesangial and capillary loop staining for IgG with corresponding subepithelial, intramembranous, and mesangial electron-dense deposits identified on electron microscopy. These are typical features of membranous lupus nephritis. #DiseaseWeek
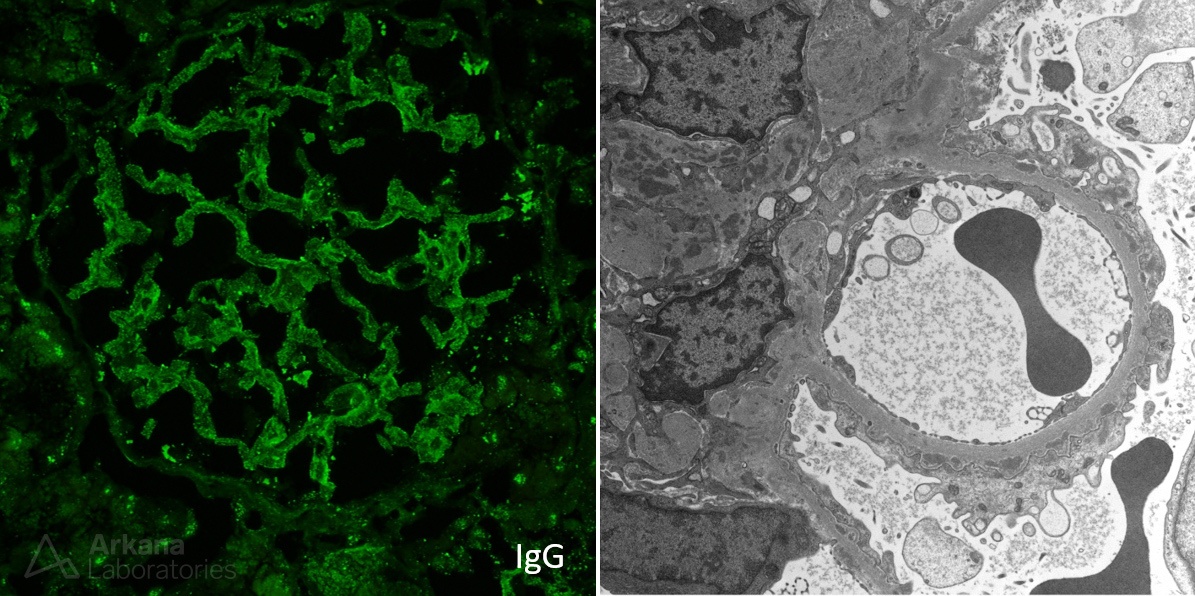
Patients with membranous lupus nephritis present with proteinuria. Unlike primary membranous glomerulopathy, there is an increased frequency of subnephrotic proteinuria, with 40% of patients having < 3 g proteinuria/day and 20% with less than 1 g/day. #DiseaseWeek
Patients with membranous lupus nephritis are less likely to show active serologies (anti-dsDNA, anti-Smith antibodies, high titer ANA), hypertension, and renal insufficiency when compared to patients with focal or diffuse lupus nephritis. #DiseaseWeek
One-third of patients with membranous lupus nephritis have isolated kidney disease prior to other systemic manifestations, which may develop later. #DiseaseWeek
Presence of these favors a membranous lupus nephritis over primary membranous:
- Mesangial deposits
- Subendothelial deposits
- C1q deposits
- “Full house” immunofluorescence
- TBM deposits
- Tissue ANA
- Tubuloreticular inclusions
- Negative PLA2R
#DiseaseWeek
In SLE patients without concurrent proliferative lupus nephritis, the differential diagnosis of membranous lupus nephritis includes other secondary causes of membranous glomerulopathy, including drug rxns, infections (including Hepatitis B + C), and malignancy. #DiseaseWeek
Vascular staining in membranous glomerulopathy indicates a secondary cause. Dr. Bourne’s teaching point at https://www.arkanalabs.com/teaching-points-6/
Dr. Best’s teaching point on subepithelial deposits in lupus nephritis https://www.arkanalabs.com/subepithelial-deposits-in-lupus-nephritis/
Laser capture microdissection studies have revealed exostosin (EXT) as an autoantigen in approximately 1/3 of cases of membranous lupus nephritis, and in fewer cases of primary idiopathic membranous glomerulopathy. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/31061139
Exostosin-1 gene mutations have been associated with familial nephropathy. Exostosin mutations impair heparan sulfate polymerization, leading to GBM depletion, disrupting the anionic charge barrier and resulting in proteinuria. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/18216313
Nephrotic syndrome in patients with SLE can be due to membranous glomerulopathy, or due to lupus podocytopathy. Lupus podocytopathy is seen in 1-2% of biopsies in patients with systemic lupus erythematosus. #DiseaseWeek
Lupus podocytopathy is diagnosed clinically by meeting clinical criteria for lupus (established by the American College of Rheumatology), the presence of nephrotic syndrome, and severe to global podocyte foot process effacement. #DiseaseWeek
Lupus podocytopathy can be seen with negative immunofluorescence, or with mesangial immune deposition, which can be accompanied by mesangial proliferation on light microscopy. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/26983708
Therefore, it can be seen without a diagnosis of lupus nephritis, with minimal mesangial lupus nephritis (class I lupus nephritis), or with mesangial proliferative lupus nephritis (class II lupus nephritis). #DiseaseWeek
The criteria for lupus podocytopathy are shown below: #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/26983708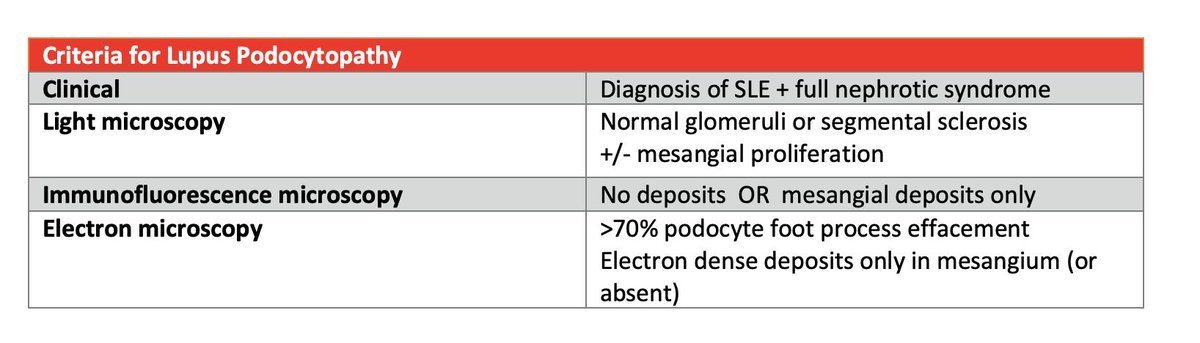
A diagnosis of minimal change disease in a lupus patient is a lupus podocytopathy. A majority of patients (94%) with lupus podocytopathy respond to immunosuppressive therapy with corticosteroids, however 59.6% relapse at 5 years. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/26983707
Collapsing glomerulopathy may be an extreme form of lupus podocytopathy, with worse outcomes. #DiseaseWeek
Collapsing glomerulopathy in SLE patients is associated with APOL1 risk alleles in African American patients. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/23520206

Answer: D. ISN/RPS class V is membranous lupus nephritis. A diagnosis of lupus podocytopathy excludes proliferative lesions (ISN/RPS class III or IV) and membranous glomerulopathy.
Thursday:
The spectrum of renal vascular lesions in lupus nephritis includes uncomplicated vascular immune deposits, lupus vasculopathy, thrombotic microangiopathy (including HUS/TTP), malignant hypertensive vascular lesions, and arteriosclerosis or arteriolosclerosis. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/9352738
The presence of renal vascular lesions predicts a poor prognosis. The 10-year survival is reduced from 83.3% (lupus nephritis without vascular lesions) to 78.8% (lupus nephritis with vascular lesions). #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/1867181
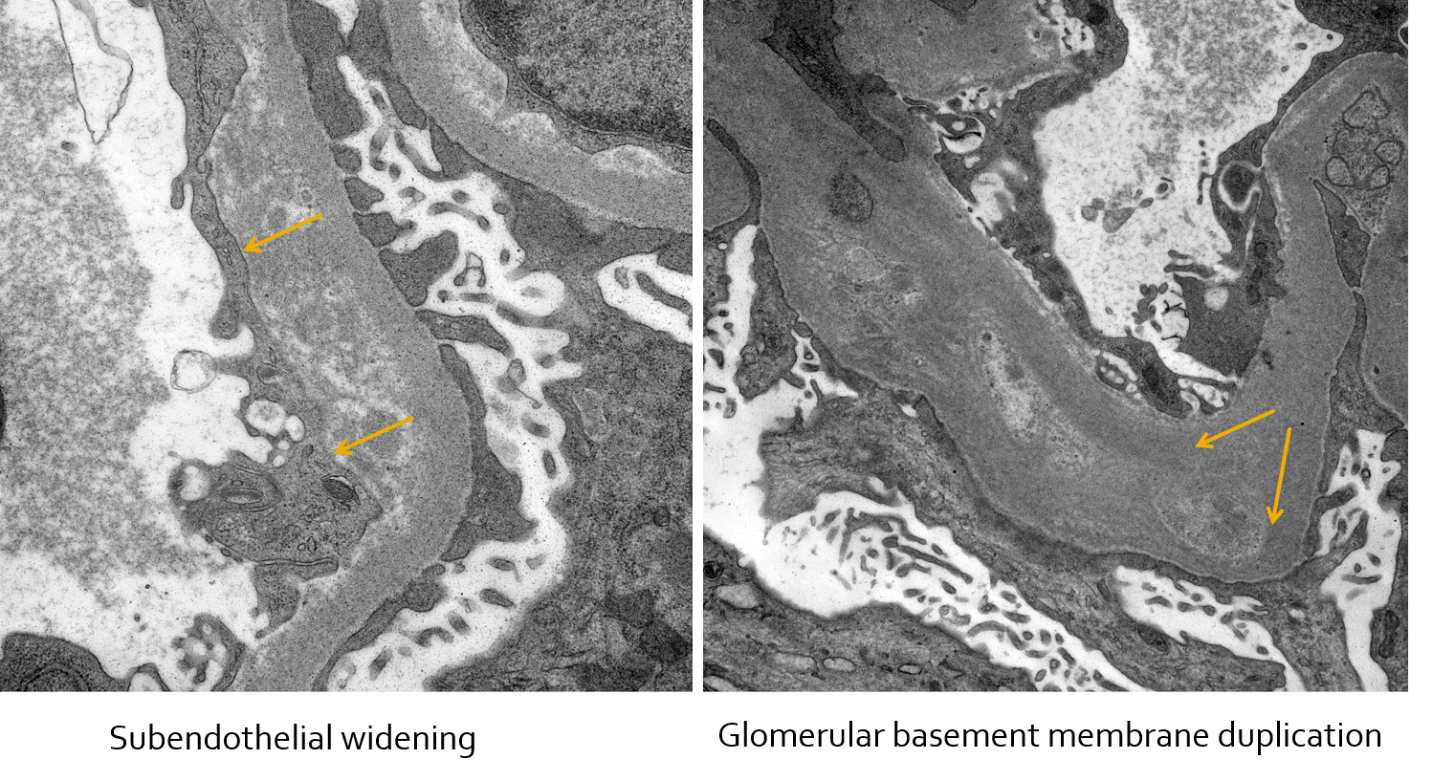
In these electron photomicrographs of a patient with lupus nephritis and chronic thrombotic microangiopathy, subendothelial widening and glomerular basement membrane duplication are shown. #DiseaseWeek
In patients with membranous lupus nephritis, similar to idiopathic membranous glomerulopathy, there is an increased risk of renal vein thrombosis and pulmonary emboli due to the hypercoagulable state of nephrotic syndrome. #DiseaseWeek
The risk of renal vein thrombosis in membranous glomerulopathy has been reported to be 5-62% in separate case series, as shown below. #DiseaseWeek
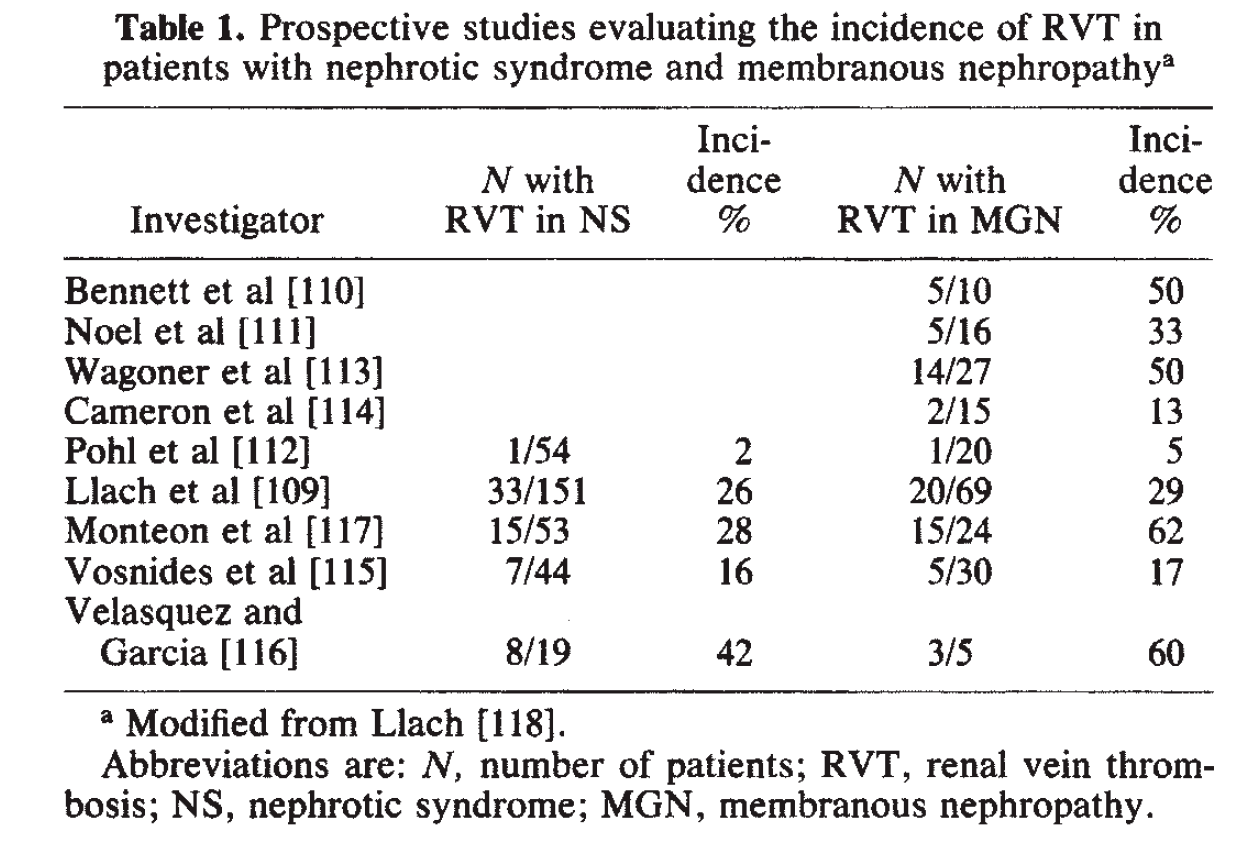
https://www.ncbi.nlm.nih.gov/pubmed/3906225
The etiology of renal vein thrombosis in nephrotic syndrome is due to urinary losses of circulating anti-coagulants, including antithrombin 3. There is increased plasma fibrinogen, clotting factors (factor V, VII, VIII, and X), and thromboplastin that promote thrombosis. Thrombocytosis is also common. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/5578557
Signs of a renal vein thrombosis on a kidney biopsy include:
- RBC congestion or neutrophil margination in glomerular capillaries
- Focal glomerular capillary loop fibrin thrombi
- Diffuse interstitial edema
- Disproportionate interstitial fibrosis and tubular atrophy to the degree of global glomerulosclerosis
12.6% of patients with SLE have positive serologies for antineutrophil cytoplasmic antibodies (ANCA). ANCA seropositivity is associated with a segmental distribution within glomeruli in diffuse lupus nephritis. #DiseaseWeek
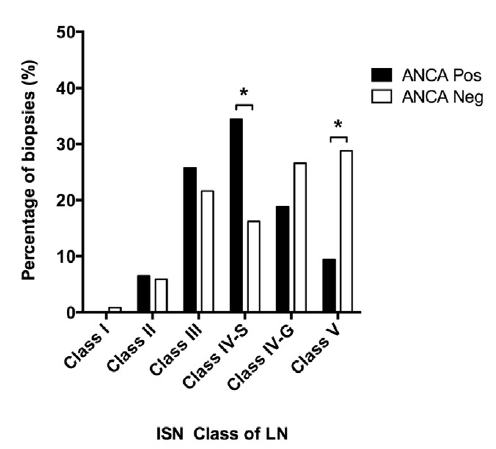
https://www.ncbi.nlm.nih.gov/pubmed/28750930
Lupus nephritis patients with a positive ANCA serology most commonly have anti-MPO antibodies (82%). Of the remaining patients, 11% had dual MPO and PR3 antibodies, and 7% had PR3 antibodies. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/28750930
Lupus nephritis with a positive ANCA serology are more likely to have active necrotizing lesions. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/28750930
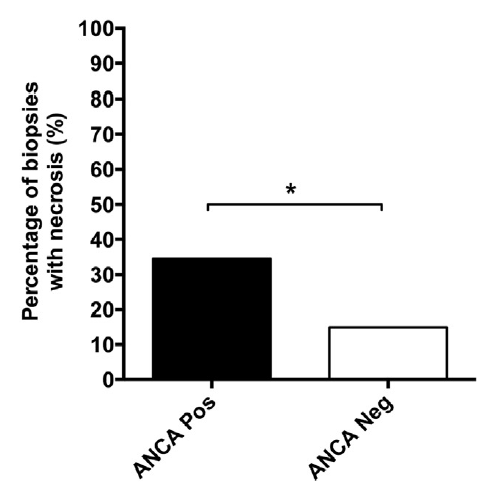
ANCA positivity in diffuse lupus nephritis is more likely to have a pauci-immune phenotype with fewer immune deposits than without ANCA positivity. Lupus patients with ANCA and class IV-S nephritis show a worse baseline renal function and have more active lupus serologies (including increased anti-dsDNA titers). #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/29055427
Antiphospholipid antibody (APL) nephropathy can present clinically with hypertension (mild, accelerated, or malignant), chronic renal failure, hematuria, +/- proteinuria. #Disease Week
APL nephropathy is associated with lupus anticoagulant, but not anticardiolipin antibodies. #DiseaseWeek
APL nephropathy presents as a spectrum of lesions within kidney biopsies, including thrombotic microangiopathy (with glomerular and vascular involvement), fibrous or mucoid intimal hyperplasia, organizing thromboses with fibrous or fibrocellular luminal stenosis, and disproportionate interstitial fibrosis and tubular atrophy due to ischemic injury. #DiseaseWeek
Up to 30% of kidney biopsies of SLE patients have APL nephropathy, with or without lupus nephritis. #DiseaseWeek
Reference: Daugas E, Nouchy D, Huong DL, et al. Antiphospholipid syndrome nephropathy in systemic lupus erythematosus. JASN 2002; 13: 42-52. https://www.ncbi.nlm.nih.gov/pubmed/11752020
APL nephropathy is associated with extrarenal arterial, but not venous, thromboses. #DiseaseWeek
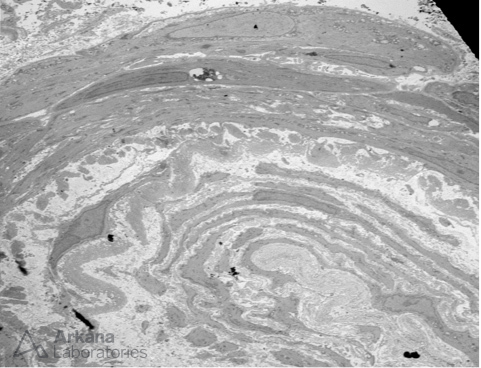
Onion-skinning like reaction due to concentric myointimal proliferation within an arteriole, seen on electron microscopy. #DiseaseWeek
Friday:
For lupus nephritis patients who develop end-stage renal disease and undergo transplantation, 2.44% have disease recurrence in the allograft kidney. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3152228/
Patients at increased risk of disease recurrence include females, age < 33 years, and non-Hispanic black race. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3152228/
Although recurrent lupus nephritis is rare, it is typically aggressive, with graft failure in 93% of patients with recurrent disease. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3152228/
Determining when to stop immunosuppression for proliferative lupus nephritis can be a difficult challenge for nephrologists and rheumatologists. A prospective observational study examined kidney biopsies following maintenance immunosuppression to address this. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/30045812
The study utilized the NIH activity and chronicity indices to evaluate for residual disease on biopsies. In patients with a residual NIH activity greater than 2, there was a risk of disease flare following cessation of immunosuppression. #DiseaseWeek
The sensitivity of a score of >2 is 100% and the specificity was 88% for flare. This study suggests a repeat kidney biopsy following immunosuppressive therapy may be helpful to determine whether it is appropriate to withdraw treatment. #DiseaseWeek https://www.ncbi.nlm.nih.gov/pubmed/30045812
Indications for re-biopsy in lupus nephritis were reviewed recently in Annals of Translational Medicine. Biopsy timing and impact on management is summarized below: #DiseaseWeek

https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6291576/
Summary:
New SLE diagnostic criteria increase sensitivity for lupus nephritis diagnosis, and novel biomarkers could prove helpful to compensate for reduced specificity.
Under the new criteria established by the Systemic Lupus International Collaborating Clinic as a joint effort from the American College of Rheumatology and European League Against Rheumatism, a diagnosis of lupus nephritis can now be made with a positive antinuclear antibody and class III or IV lupus nephritis alone. This increased the sensitivity of diagnosis but reduced specificity, when compared to the 1997 revision of the 1982 American College of Rheumatology SLE diagnostic criteria (Rijnink et al, 2018). With the current classification, lupus-like conditions, such as HIV-associated immune complex disease of the kidney (HIVICK) could be included in a diagnosis of SLE. The development of biomarkers are needed to differentiate lupus nephritis from other lupus-like conditions, such as infections.
The ISN/RPS classification is used to evaluate lupus nephritis on a kidney biopsy. The NIH activity and chronicity indices provide comparisons of disease progression and response to therapy.
The International Society of Nephrology/ Renal Pathology Society classification separates categories of lupus nephritis, but does not provide prognostic value. The NIH Disease Activity and Chronicity indices is used in parallel to do this, but at present, there are no clear cutoffs of activity or chronicity that indicate when to treat lupus nephritis patients (Bajema et al, 2018). Establishing disease activity (and/or chronicity) cutoffs to guide when to start immunosuppressive therapy will be important. There are also likely to be subcategories of proliferative lupus nephritis that may have clinical and prognostic significance. One example are the segmental necrotizing lesions in patients with focal or diffuse lupus nephritis in patients with concurrent positive ANCA serology that predict poorer renal outcomes. The classification of lupus nephritis is also limited by glomerulocentricity, since severe tubulointerstitial injury and vascular involvement have a poor renal prognosis but are not included in the classification scheme.
A lupus classification based on the antigenic drivers of disease may exist in the future.
It is possible that lupus nephritis may be classified according to the inciting autoantigen in the future. Laser capture microdissection studies and elution of immune complexes from tissue combined with proteonomic techniques can be used to identify autoantigens within immune complex-mediated diseases. One recent example is the exostosin 1/2 complex, which was recently identified in membranous lupus nephritis (Sethi et al, 2019). There are likely several other autoantigenic drivers that have yet to be identified and could represent distinct forms of the disease. Development of serological assays could allow for monitoring of disease progression and response to therapy, as is available with the two current antigens for idiopathic membranous glomerulopathy (PLA2R and THSD7A).
Improved serum and urine biomarkers could have predictive value and inform treatment.
Non-invasive serum and urine biomarkers are needed to predict the onset of lupus nephritis in patients with SLE and to predict flares for patients with established disease. Currently, SLE patients are monitored by nephrologists and rheumatologists by non-specific laboratory tests, including serum creatinine, estimated glomerular filtration rate, and urine studies (urine sediment analysis, proteinuria, and hematuria), serum complements, and serologic studies. Anti-dsDNA titers, if present, can be helpful to predict flare, as well as low serum complements. Proteinuria at one year is the best predictor, at present, for long-term kidney outcome. However, resolution of serum creatinine and reduction of proteinuria does not always correlate with the absence of disease activity. Kidney biopsy studies have shown that residual histologic activity is present despite improved serum creatinine and/or proteinuria. Due to this, repeat kidney biopsies may be useful post-treatment to determine when to stop immunosuppressive therapy. An NIH disease activity score >2 on a post-treatment biopsy indicates poor outcomes.
There is a need for development and validation for serum and urine biomarkers to predict disease flare and response to therapy. Prospective studies evaluating novel analytes in clinical trials with long-time follow-up will be helpful to stratify risk, predict flare, monitor response to therapy, and predict disease prognosis. Molecular analyses of kidney biopsies could also be helpful in this process (Parikh et al, 2015)
Enrollment in clinical trials may establish new treatment targets and provide hope to patients at risk of renal failure.
Some factors associated with poor outcomes in lupus nephritis include African or Hispanic ethnicity, male sex, earlier age of onset, frequent disease relapses, incomplete remissions post-immunosuppressive therapy, proteinuria >4 grams at diagnosis, presence of anti-phospholipid antibodies, high titer dsDNA antibodies, anti-C1q antibodies, and crescentic disease or vascular involvement on a kidney biopsy (Rovin et al, 2019). Optimal management for many of these at risk patients has yet to be determined. One example of improved treatment in an at-risk group is mycophenolate mofetil for proliferative lupus nephritis, which has shown improved outcomes in African American patients. Future studies are needed to determine how to optimally treat patients in these risk groups. Increased identification and enrollment in clinical trials will help evaluate the efficacy of new targets and to optimize therapy.
References:
Bajema IM, Wihelmus S, Alpers CE, et al. Revision of the International Society Classification for Lupus Nephritis: Clarification of Definitions, and Modified National Institutes of Health Activity and Chronicity Indices. Kidney International 2018; 93: 789-796.
Parikh SV, Malvar A, Song H, et al. Characterizing the Immune Profile of the Kidney Biopsy at Lupus Nephritis Flare Differentiates Early Treatment Responders from Non-Responders. Lupus Sci Med 2015; 2: e000112.
Rijnink EC, Teng YKO, Kraaji T, et al. Validation of the Systemic Lupus International Collaborating Clinics Classification Criteria in a Cohort of Patients with Full House Glomerular Deposits. Kidney International 2018; 93: 214-220.
Rovin BH, Caster DJ, Cattran DC, Gibson KL, Hogan JJ, Moeller MJ, Roccatello D, Cheung M, Wheeler DC, Winkelmayer WC, Floege J. Management and Treatment of Glomerular Diseases (Part 2): Conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference. Kidney International 2019; 95: 281-295.
Sethi S, Madden BJ, Debiec H, Charlesworth MC, Gross L, Ravidran A, Hummel AM, Specks U, Fervenza FC, Ronco P. Exostosin 1/exostosin 2-associated membranous nephropathy. JASN 2019 Jun 30 (6): 1123-1136
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.




