This 76-year-old white female developed nephrotic syndrome over about 3 months. She has proteinuria, 9 gm/24h and an elevated creatinine of 2.1 mg/dl (baseline 1.9 mg/dl 6 months prior to biopsy). Urinalysis showed 3+ protein and 2+ blood but no casts. ANA, ANCA and complement levels are unremarkable.
Legend:
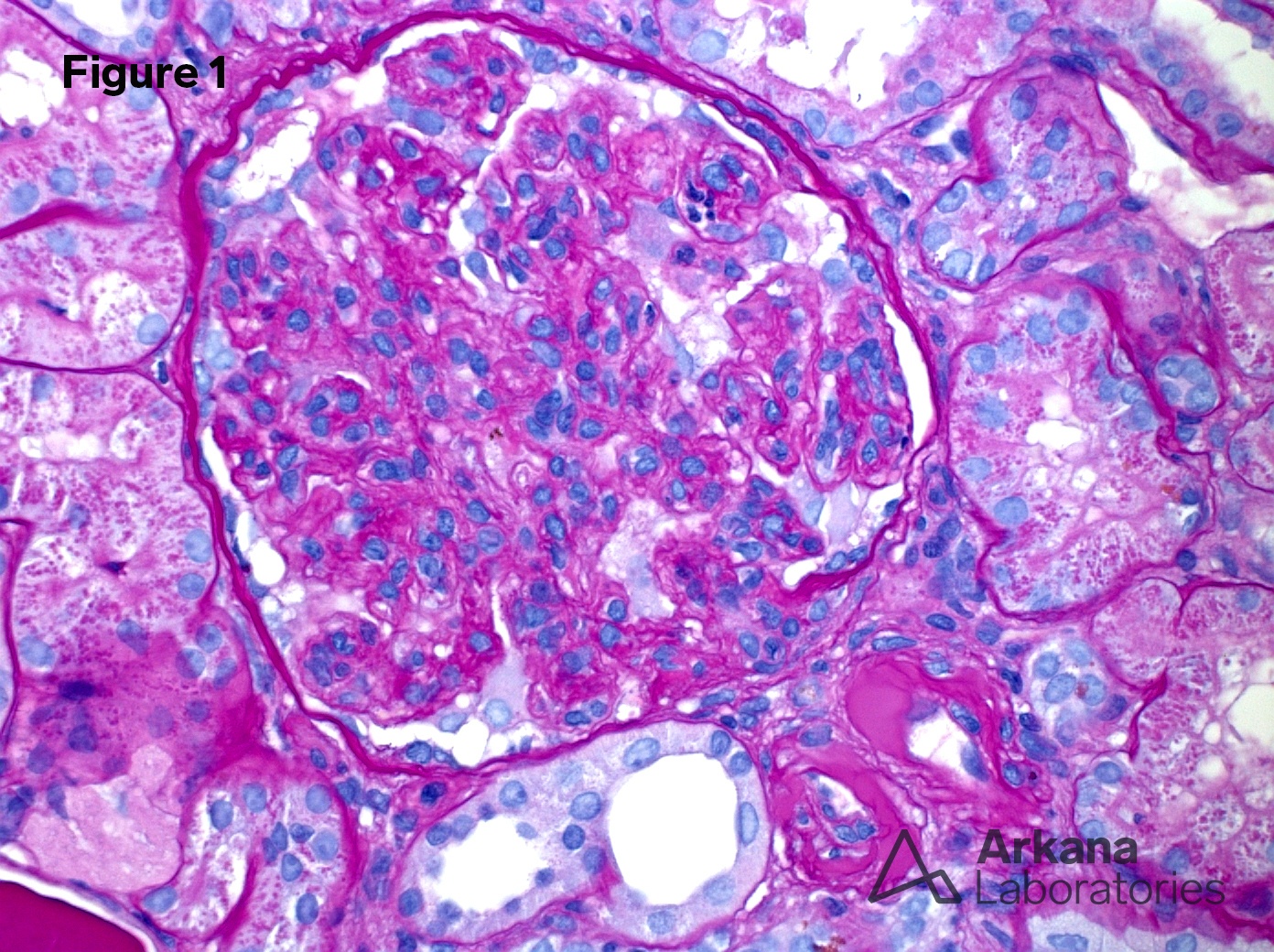
1. Mesangial and endocapillary proliferation (PAS x400)
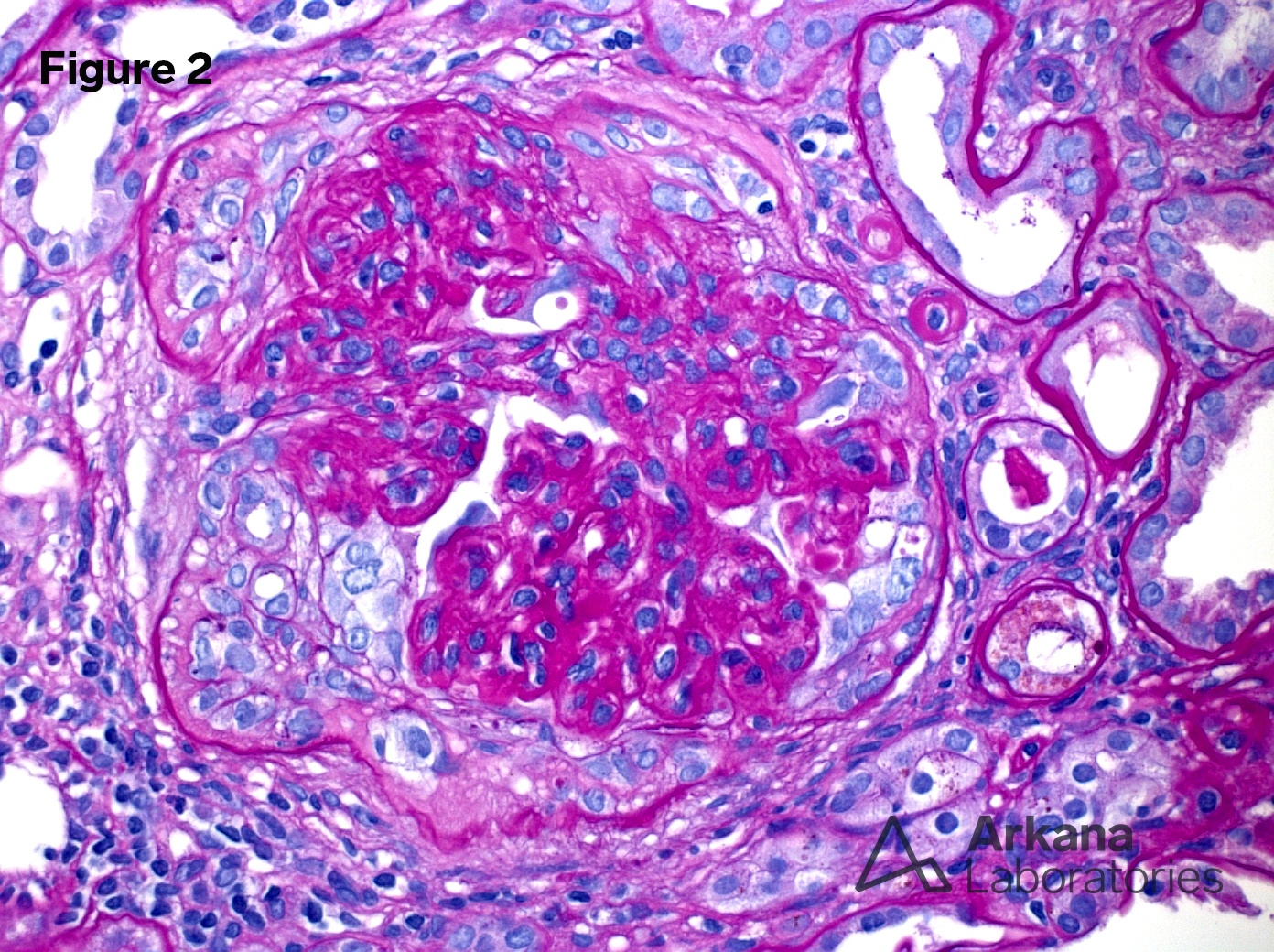
2. Crescent (PAS x400)
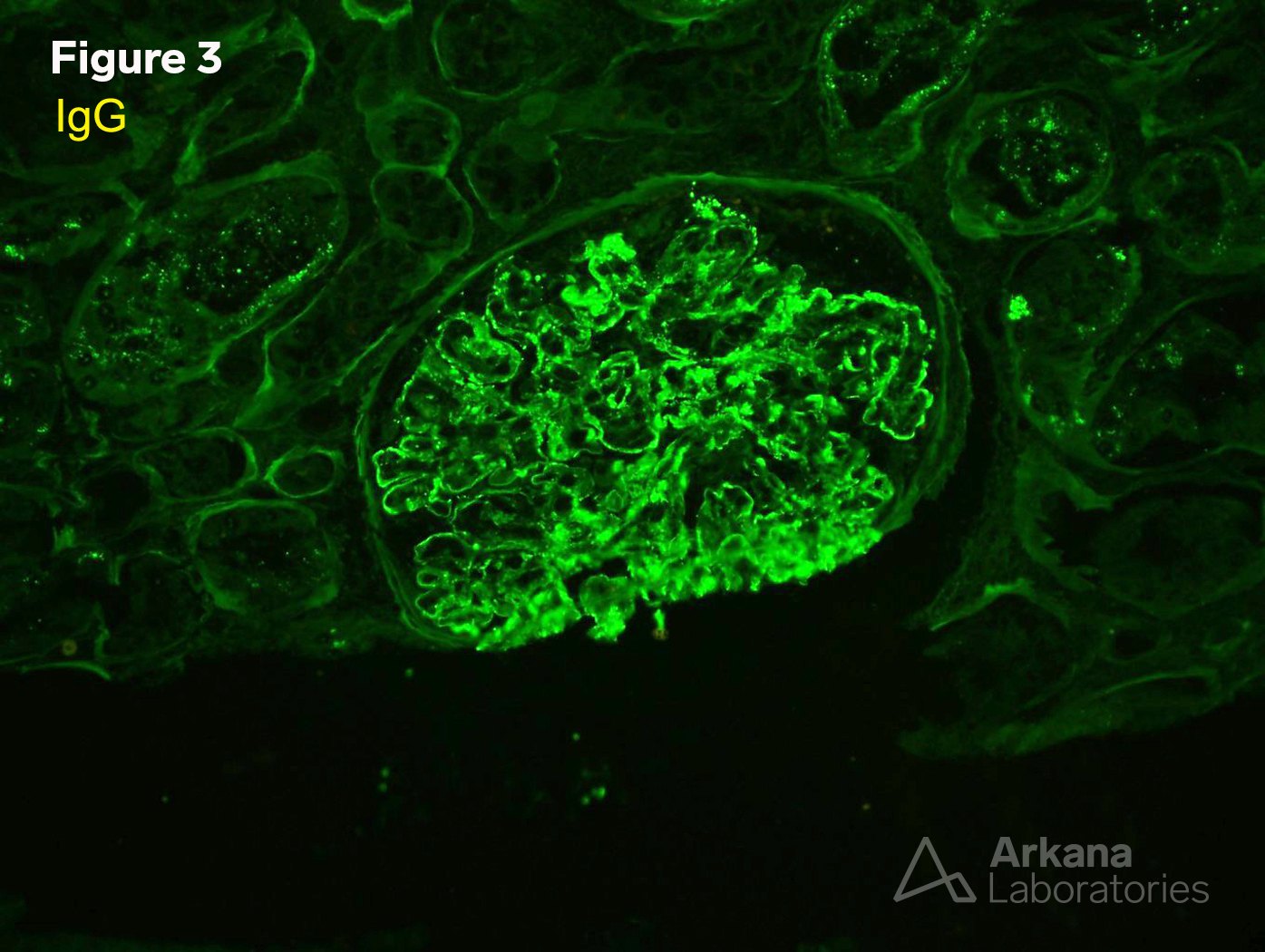
3. IgG (IF x200)
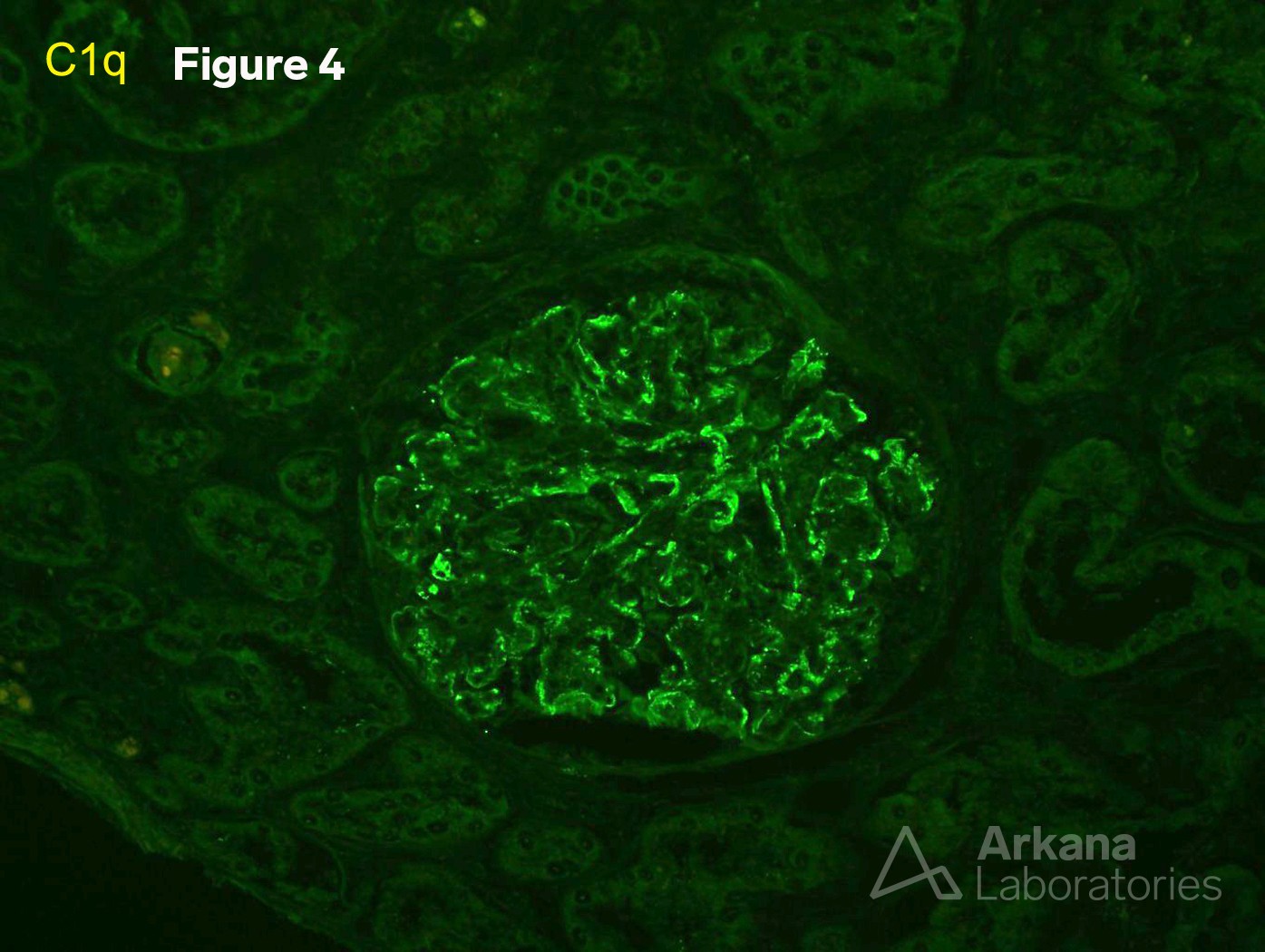
4. C1q (IF x200.)
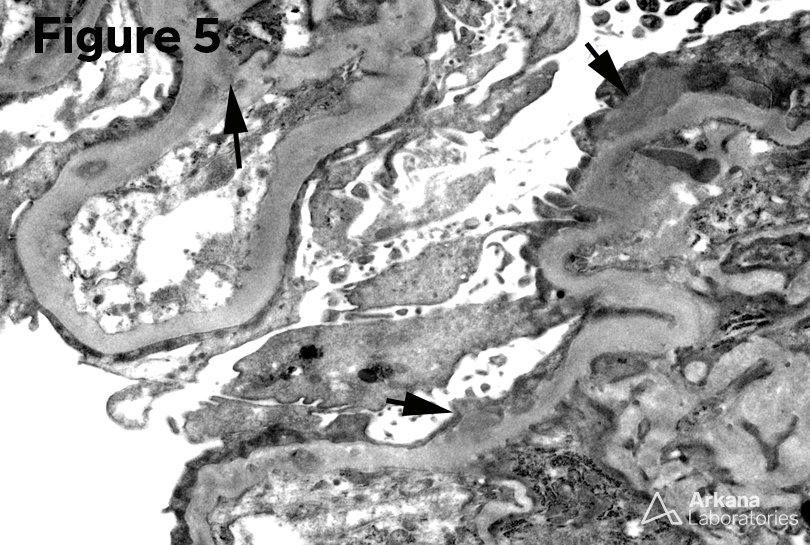
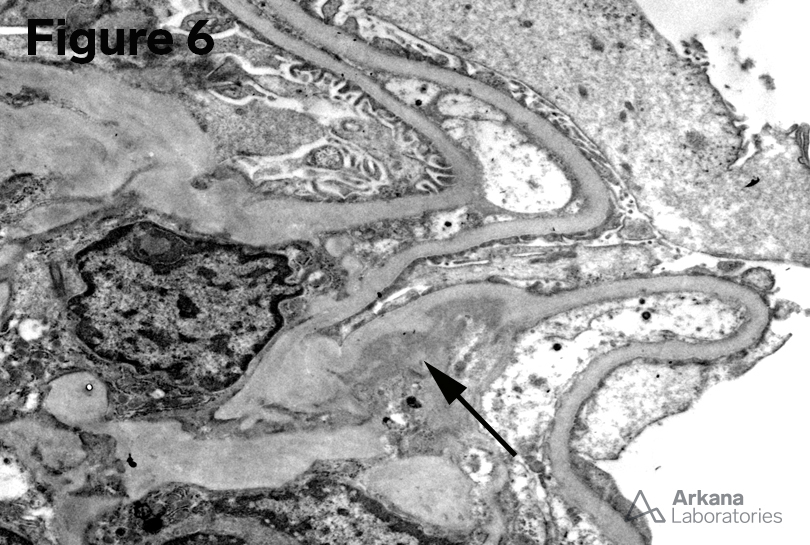
5. Mesangial and Loop deposits (EM x6000)
6. Mesangial deposits (EM x6000)
Diagnosis:
Proliferative and crescentic glomerulonephritis with monoclonal immunoglobulin deposits.
Comment: IgG subtyping revealed monoclonal IgG type 3.
Proliferative glomerulonephritis with monoclonal immunoglobulin deposits (PGMID) is a diagnostic mouthful describing a unique syndrome that occurs predominantly in white (>85%) women (>60%) older than 50 (mean age 55 years) presenting with nephrotic syndrome (50%), renal insufficiency (70%) and hematuria (>75%) (1). 30% had an MGUS but only one patient was found to have myeloma. After 30 months follow up, 40% were in remission, 40% were persistently symptomatic and 20% had progressed to end-stage renal failure. In small series, rituximab has been shown to have a positive impact on PGMID (2).
References:
[1] Nasr SH, Satoskar A, Markowitz G, Valeri A, Appel GB, Stokes MB, Nadasdy T, D’Agati VD: Proliferative glomerulonephritis with monoclonal IgG deposits. J Am Soc Nephrol 2009, 20:2055-64.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2736767/
[2] Guiard E, Karras A, Plaisier E, Duong Van Huyen JP, Fakhouri F, Rougier JP, Noel LH, Callard P, Delahousse M, Ronco P: Patterns of noncryoglobulinemic glomerulonephritis with monoclonal Ig deposits: correlation with IgG subclass and response to rituximab. Clin J Am Soc Nephrol 2011, 6:1609-16.
http://cjasn.asnjournals.org/content/6/7/1609.long
Quick note: This post is to be used for informational purposes only and does not constitute medical or health advice. Each person should consult their own doctor with respect to matters referenced. Arkana Laboratories assumes no liability for actions taken in reliance upon the information contained herein.